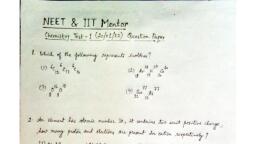Question 1 :
A container has hydrogen and oxygen mixture in ratio of 1 : 1 by weight, then
Question 3 :
All the naturally occurring processes, <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7d29fab3481716f4b76e7"> spontaneous proceed spontaneously in a direction which leads to:
Question 4 :
A closed flask contains water in all its three states, solids, liquid and vapour at <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7d1cd399925718ac6afbd"> In this situation the average KE of the water molecule will be:
Question 5 :
At constant pressure and temperature, the direction of any chemical reaction is one where, the decrease.
Question 6 :
The exchange of heat energy during chemical reaction at constant temperature and pressure occurs in form of:
Question 8 :
When a gas undergoes adiabatic expansion, it gets cooled due to
Question 10 :
For the reaction of one mole of zinc dust with one mole of sulphuric acid in a bomb calorimeter, {tex} \Delta \mathrm { U } {/tex} and {tex} \mathrm { w } {/tex} corresponds to
Question 11 :
Standard enthalpy of combustion of {tex} \mathrm { CH } _ { 4 } {/tex} is {tex} - 890 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex} and standard enthalpy of vaporisation of water is {tex} 40.5 \mathrm { kJ } \mathrm { mol } ^ { - 1 } . {/tex} The enthalpy change of the reaction<br>{tex} \mathrm { CH } _ { 4 } ( \mathrm { g } ) + 2 \mathrm { O } _ { 2 } ( \mathrm { g } ) \longrightarrow \mathrm { CO } _ { 2 } ( \mathrm { g } ) + \mathrm { H } _ { 2 } \mathrm { O } ( \mathrm { g } ) {/tex}<br>
Question 12 :
The work done during the expansion of a gas from a volume of 4{tex}\ \mathrm { dm } ^ { 3 } {/tex} to 6{tex}\ \mathrm { dm } ^ { 3 } {/tex} against a constant external pressure of 3{tex}\ \mathrm { atm } {/tex} is {tex} ( 1\ \mathrm { L }\ \mathrm { atm } = 101.32\ \mathrm { J } ) {/tex}
Question 14 :
Consider the following reactions:<br> {tex}\mathrm {(i)}\ \mathrm{ H^+}_{(aq)} + \mathrm{ OH^-}_{(aq)} = \mathrm{ H_2O_{(l)}}, \ \Delta H = -X_1\ \mathrm {{kJ}\ mol^{-1}} {/tex}<br> {tex}\mathrm {(ii)}\ \mathrm{ H}_{2(g)} + \mathrm{ 1/2 O}_{2(g)} = \mathrm{ H_2O_{(l)}}, \ \Delta H = -X_2\ \mathrm {{kJ}\ mol^{-1}} {/tex} <br>{tex}\mathrm {(iii)}\ \mathrm{ CO}_{2(g)} + \mathrm{ H}_{2(g)} = \mathrm{ CO_{(g)} + \mathrm{ H_2O_{(l)}}}, \ \Delta H = -X_3\ \mathrm {{kJ}\ mol^{-1}} {/tex} <br>{tex}\mathrm {(iv)}\ \mathrm{C_2 H}_{2(g)} + \mathrm{ 5/2O}_{2(g)} = \mathrm{2CO_{2(g)} + \mathrm{ H_2O_{(l)}}}, \ \Delta H = +X_4\ \mathrm {{kJ}\ mol^{-1}} {/tex} <br> Enthalpy of formation of {tex}\mathrm {H_2O}_{(l)}{/tex} is
Question 15 :
One mole of a non-ideal gas undergoes a change of state {tex} ( 2.0 \text { atm, } 3.0 \mathrm { L } , 95 \mathrm { K } ) \rightarrow ( 4.0 \mathrm { atm } , 5.0 \mathrm { L } , 245 \mathrm { K } ) {/tex} with a change in internal energy, {tex} \Delta \mathrm { U } = 30.0 \mathrm { Latm } . {/tex} The change in enthalpy {tex} \Delta \mathrm { H } {/tex} of the process in {tex} \mathrm { L } {/tex} atm is.
Question 16 :
The enthalpy of neutralisation of a weak acid in {tex} 1 \mathrm { M } {/tex} solution with a strong base is {tex} - 56.1 \mathrm { kcal } {/tex} {tex} \mathrm { mol } ^ { - 1 } {/tex}. If the enthalpy of ionisation of acid is {tex} 1.5 \mathrm { kcal } \mathrm { mol } ^ { - 1 } {/tex} and enthalpy of neutralisation of the strong acid with a strong base is {tex} - {/tex} {tex} 57.3 \mathrm { kJ } \ \mathrm { eq } ^ { - 1 } . {/tex} What is the % ionisation of the weak acid in molar solution (assume the acid is monobasic)
Question 18 :
The free energy change for the following reactions are given below,<br> {tex} \mathrm { C } _ { 2 } \mathrm { H } _ { 2 } ( \mathrm { g } ) + \frac { 5 } { 2 } \mathrm { O } _ { 2 } ( \mathrm { g } ) \rightarrow {/tex}{tex} 2 \mathrm { CO } _ { 2 } ( \mathrm { g } ) \rightarrow \mathrm { CO } _ { 2 } ( \mathrm { g } ) ; \Delta \mathrm { G } ^ { \circ } = - 1234 \mathrm { kJ }{/tex} <br> {tex} \mathrm { C } ( \mathrm { g } ) + \mathrm { O } _ { 2 } ( \mathrm { g } ) \rightarrow \mathrm { CO } _ { 2 } ( \mathrm { g } ) ; \Delta \mathrm { G } ^ { \circ } = - 394 \mathrm { kJ } {/tex}<br> {tex} \mathrm { H } _ { 2 } ( \mathrm { g } ) + \frac { 1 } { 2 } \mathrm { O } _ { 2 } ( \mathrm { g } ) \rightarrow \mathrm { H } _ { 2 } \mathrm { O } ( \mathrm { l } ) ; \Delta \mathrm { G } ^ { \circ } = - 237 \mathrm { kJ } {/tex} <br>What is the standard free energy change for the reaction<br> {tex} \mathrm { H } _ { 2 } ( \mathrm { g } ) + 2 \mathrm { C } ( \mathrm { s } ) \longrightarrow \mathrm { C } _ { 2 } \mathrm { H } _ { 2 } ( \mathrm { g } ) {/tex}
Question 19 :
If the enthalpy change for the transition of liquid water to steam is 30kJ {tex}mol^{-1}{/tex} at {tex}27^\circ{/tex} C, the entropy change for the proces would be
Question 20 :
Standard entropies of {tex}\mathrm X_2, \mathrm Y_2{/tex} and {tex} \mathrm {XY_3 } {/tex} are {tex}60, 40{/tex} and {tex}50{/tex} {tex}\mathrm{J K}{/tex} {tex}^{-1} {/tex} {tex}\mathrm {mol^{-1}} {/tex} respectively. For the reaction {tex}1/2\mathrm X_2 + 3/2\mathrm Y_2 \rightleftharpoons \mathrm {XY}_3, \Delta H = -\mathrm {30\ kJ} {/tex} to be at equilibrium, the temperature should be
Question 21 :
Following reaction occurring in an automobile <br>{tex} 2 \mathrm { C } _ { 8 } \mathrm { H } _ { 18 ( g ) } + 25 \mathrm { O } _ { 2 ( g ) } \rightarrow 16 \mathrm { CO } _ { 2 ( g ) } + 18 \mathrm { H } _ { 2 } \mathrm { O } _ { ( g ) } {/tex}<br> The sign of {tex} \Delta H , \Delta S {/tex} and {tex} \Delta G {/tex} would be
Question 22 :
{tex}\mathrm{2Zn\ +\ O_2\ \rightarrow\ 2ZnO;\ \Delta}G^\circ = -616 \ \mathrm J {/tex}<br>{tex}\mathrm{2Zn\ +\ S_2\ \rightarrow\ 2ZnS;\ \Delta}G^\circ = -293 \ \mathrm J {/tex}<br>{tex}\mathrm{S_2\ +\ 2O_2\ \rightarrow\ 2SO_2;\ \Delta}G^\circ = -408 \ \mathrm J {/tex}<br>{tex}\Delta G^\circ {/tex} for the following reaction<br>{tex}\mathrm{2ZnS\ +\ 3O_2\ \rightarrow\ 2ZnO\ +\ 2SO_2} {/tex} is
Question 23 :
Heat of combustion {tex} \Delta H {/tex} for {tex} C _ { ( s ) } , H _ { 2 ( g ) } {/tex} and {tex} C H _ { 4 ( g ) } {/tex} are {tex} - 94 , - 68 {/tex} and {tex} - 213 {/tex} kcal/mol, then {tex} \Delta H {/tex} for {tex} C _ { ( s ) } + 2 H _ { 2 ( g ) } \rightarrow C H _ { 4 ( g ) } {/tex} is
Question 24 :
The enthalpy of atomisation of {tex} \mathrm { CH } _ { 4 } {/tex} and {tex} \mathrm { C } _ { 2 } \mathrm { H } _ { 6 } {/tex} are {tex}360{/tex} and {tex} 620 \ \mathrm { kcal } \mathrm { mol } ^ { - 1 } {/tex} respectively. The {tex} \mathrm { C } - \mathrm { C } - \mathrm { C } {/tex} bond energy is expected to be
Question 25 :
Combustion of sucrose is used by aerobic organisms for providing energy for the life sustaining processes. If all the capturing of energy from the reaction is done through electrical process (non {tex} \mathrm { P-V } {/tex} work) then calculate maximum available energy which can be captured by combustion of {tex}34.2 g{/tex} of sucrose Given: {tex}\mathrm {\Delta H_{combustion}(sucrose)} = 6000 \mathrm { kJ mol}^{-1} {/tex} Given : {tex}\mathrm {\Delta S_{combustion}} = \mathrm {180 J/K mol }{/tex} and body temperature is 300 K.
Question 26 :
The standard enthalpies of formation of {tex} \mathrm { CO } _ { 2 } ( \mathrm { g } ) , \mathrm { H } _ { 2 } \mathrm { O } ( \mathrm { I } ) {/tex} and glucose(s) at {tex} 25 ^ { \circ } \mathrm { C } {/tex} are {tex} - 400 \mathrm { kJ } / \mathrm { mol } , - 300 \mathrm { kJ } / \mathrm { mol } {/tex} and {tex} - 1300 {/tex} {tex} \mathrm { kJ } / \mathrm { mol } , {/tex} respectively. The standard enthalpy of combustion of glucose at {tex} 25 ^ { \circ } \mathrm { C } {/tex} is
Question 27 :
Equal volumes of methanoic acid and sodium hydroxide are mixed. If {tex}\mathrm x {/tex} is the heat of formation of water, then heat evolved on neutralization is
Question 28 :
Match the columns<br>
<table>
<tr><th>Column I </th> <th>Column II </th> </tr>
<tr><td>A. {tex}\mathrm {C_4H_{10}+\frac{13}{2}O_2\rightarrow4CO_2+5H_2O};\Delta H=-w {/tex}</td> <td>I.Enthalpy of atomisation</td> </tr>
<tr><td>B.{tex}\mathrm {CH_4\rightarrow C+4H}; \Delta H= x{/tex}</td> <td>II.Enthalpy of formation</td> </tr>
<tr><td>C. {tex}\mathrm {H_2+Br_2\rightarrow 2HBr};\Delta H= y{/tex}</td> <td>III.Enthalpy of combustion</td> </tr>
<tr><td>D. {tex}\mathrm {Na^-(s)\rightarrow Na(g);
\Delta H= z}{/tex}</td> <td>IV. Enthalpy of sublimation</td> </tr>
</table>
Question 29 :
A certain reaction is non spontaneous at {tex} 298 \mathrm { K } {/tex}. The entropy change during the reaction is {tex} 121 \mathrm { JK } ^ { - 1 } {/tex}. If the reaction is endothermic or exothermic, the minimum value of {tex} \Delta \mathrm { H } {/tex} for the reaction is
Question 30 :
What is the value of change in internal energy at 1 atm in the process?<br>{tex} \mathrm { H } _ { 2 } \mathrm { O } ( 1,323 \mathrm { K } ) \longrightarrow \mathrm { H } _ { 2 } \mathrm { O } ( \mathrm { g } , 423 \mathrm { K } ) {/tex} <br>Given : {tex} \mathrm { C } _ { \mathrm { v } , \mathrm { m } } \left( \mathrm { H } _ { 2 } \mathrm { O } , \mathrm { l } \right) = 75.0 \mathrm { JK } ^ { - 1 } \mathrm { mol } ^ { - 1 } {/tex}<br> {tex} \mathrm { C } _ { \mathrm { p } , \mathrm { m } } \left( \mathrm { H } _ { 2 } \mathrm { O } , \mathrm { g } \right) = 33.314 \mathrm { JK } ^ { - 1 } \mathrm { mol } ^ { - 1 } {/tex}<br> {tex} \Delta \mathrm { H } _ { \mathrm { vap } } {/tex} at {tex} 373 \mathrm { K } = 40.7 \mathrm { kJ } / \mathrm { mol } {/tex}<br>
Question 31 :
The densities of graphite and diamond at 298{tex}\ \mathrm { K } {/tex} are 2.25 and 3.31{tex}\ \mathrm { g }\ \mathrm { cm } ^ { - 3 } , {/tex}respectively. If the standard free energy difference {tex} \left( \Delta G ^ { \circ } \right) {/tex} is equal to 1895{tex}\ \mathrm { J }\ \mathrm { mol } ^ { - 1 } {/tex}, the pressure at which graphite will be transformed into diamond at 298{tex}\ \mathrm { K } {/tex} is
Question 33 :
When {tex}1{/tex} mole of oxalic acid is treated with excess of {tex} \mathrm { NaOH } {/tex} in dilute aqueous solution, {tex} 106 \mathrm { kJ } {/tex} of heat is liberated. Predict the enthalpy of ionisation of the acid
Question 34 :
Using the data provided, calculate the multiple bond energy {tex} \left( \mathrm { k } \mathrm { J } \mathrm { mol } ^ { - 1 } \right) {/tex} of a {tex} \mathrm { C } \equiv \mathrm { C } {/tex} bond in {tex} \mathrm { C } _ { 2 } \mathrm { H } _ { 2 } . {/tex} That energy is (take the bond energy of a {tex}\mathrm { C } - \mathrm { H } {/tex} bond as {tex} 350 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex} )<br>{tex} 2 \mathrm { C } ( s ) + \mathrm { H } _ { 2 } ( g ) \longrightarrow \mathrm { HC } \equiv \mathrm { CH } ( g ) ;\ \Delta \mathrm { H } = 225 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex}<br> {tex} 2 \mathrm { C } ( s ) \longrightarrow 2 \mathrm { C } ( g ) ;\ \Delta \mathrm { H } = 1410 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex}<br> {tex} \mathrm { H } _ { 2 } ( g ) \longrightarrow 2 \mathrm { H } ( g ) ;\ \Delta \mathrm { H } = 330 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex}<br>
Question 35 :
Given that:<br> (i) {tex} \Delta _ { \mathrm { f } } \mathrm { H } ^ { \circ } {/tex} of {tex} \mathrm { N } _ { 2 } \mathrm { O } {/tex} is {tex} 82 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex}<br> (ii) Bond energies of {tex} \mathrm { N } \equiv \mathrm { N } , \mathrm { N } = \mathrm { N } , \mathrm { O } = \mathrm { O } {/tex} and {tex} \mathrm { N } = \mathrm { O } {/tex} are {tex} 946,418,498 {/tex} and {tex} 607 \mathrm { kJ } {/tex} {tex} \mathrm { mol } ^ { - 1 } {/tex} respectively, <br>The resonance energy of {tex} \mathrm { N } _ { 2 } \mathrm { O } {/tex} is:
Question 36 :
For the reaction taking place at certain temperature {tex} \mathrm { NH } _ { 2 } \mathrm { COONH } _ { 4 } ( \mathrm { s } ) \rightleftharpoons 2 \mathrm { NH } _ { 3 } ( \mathrm { g } ) + \mathrm { CO } _ { 2 } ( \mathrm { g } ) {/tex} if equilibrium pressure is {tex} 3 \mathrm { X } {/tex} bar then {tex} \Delta _ { \mathrm { r } } \mathrm { G } ^ { \circ } {/tex} would be
Question 37 :
The difference between heats of reaction at constant pressure and constant volume for the reaction :<br>{tex} 2 \mathrm { C } _ { 6 } \mathrm { H } _ { 6 } ( \mathrm { l } ) + 15 \mathrm { O } _ { 2 } ( \mathrm { g } ) \rightarrow 12 \mathrm { CO } _ { 2 } ( \mathrm { g } ) + 6 \mathrm { H } _ { 2 } \mathrm { O } ( \mathrm { l } ) {/tex} at {tex} 25 ^ { \circ } \mathrm { C } {/tex} in {tex} \mathrm { kJ } {/tex} is<br>
Question 38 :
For the combustion reaction at {tex} 298 \mathrm { K } {/tex} {tex} 2 \mathrm { Ag } ( \mathrm { s } ) + 1 / 2 \mathrm { O } _ { 2 } ( \mathrm { g } ) \longrightarrow 2 \mathrm { Ag } _ { 2 } \mathrm { O } ( \mathrm { s } ) {/tex}<br> Which of the following alternatives is correct?
Question 39 :
The following two reactions are known:<br>{tex} \mathrm { Fe } _ { 2 } \mathrm { O } _ { 3 } ( s ) + 3 \mathrm { CO } ( g ) \longrightarrow 2 \mathrm { Fe } ( s ) + 3 \mathrm { CO } _ { 2 } ( g ) ; \Delta \mathrm { H } = - 26.8 \mathrm { kJ } {/tex} {tex} \mathrm { FeO } ( s ) + \mathrm { CO } ( g ) \longrightarrow \mathrm { Fe } ( s ) + \mathrm { CO } _ { 2 } ( g ) ; \Delta \mathrm { H } = - 16.5 \mathrm { kJ } {/tex}<br>The value of {tex} \Delta \mathrm { H } {/tex} for the following reaction {tex} \mathrm { Fe } _ { 2 } \mathrm { O } _ { 3 } ( s ) + \mathrm { CO } ( g ) \longrightarrow 2 \mathrm { FeO } ( s ) + \mathrm { CO } _ { 2 } ( g ) \mathrm { is } {/tex}
Question 40 :
The standard enthalpy of formation of {tex} \mathrm { NH } _ { 3 } {/tex} is {tex} - 46.0 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex}. If the enthalpy of formation of {tex} \mathrm { H } _ { 2 } {/tex} from its atoms is {tex} - 436 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex} and that of {tex} \mathrm { N } _ { 2 }{/tex} is {tex}- 712 \mathrm { kJ } \mathrm { mol } ^ { - 1 } , {/tex} the average bond enthalpy of {tex} \mathrm { N } - \mathrm { H } {/tex} bond in {tex} \mathrm { NH } _ { 3 } {/tex} is