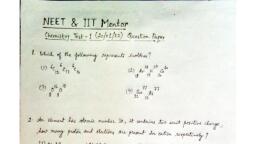Question 1 :
The oxidation state of Barium in $Ba({ H }_{ 2 }{ PO }_{ 2 }{ ) }_{ 2 }$  is:
Question 2 :
$0.7\ g$ of $Na_{2}CO_{3}. xH_{2}O$ is dissolved in $100\ ml, 20\ ml$ of which required $19.8\ ml$ of $0.1\ N\ HCl$. The value of $x$ is:
Question 3 :
Assertion: Transition metals show variable valence.
Reason: Due to a large energy difference between the $ ns^2 $ and $ ( n - 1)d $ electrons.
Question 4 :
Among cellulose, polyvinyl chloride, nylon and natural rubber, in which of the polymer intermolecular force of attraction is weakest?
Question 7 :
Assertion: Sodium chloride ($NaCl$) is a stable ionic solid.
Reason: $NaCl$ has high lattice enthalpy.
Question 8 :
{tex} \mathrm { CaO } {/tex} and {tex} \mathrm { NaCl } {/tex} have the same crystal structure and approximately the same ionic radii. If {tex}\mathrm U {/tex} is the lattice energy of {tex} \mathrm { NaCl } {/tex}, the approximate lattice energy of {tex} \mathrm { CaO } {/tex} is
Question 10 :
The conductivity of the metal decreases with increase in temperature because
Question 11 :
Which of the following is not a property of an ionic compound?
Question 12 :
Which of the following is/are misconception(s) associated with resonance?<br>(i) The molecule exists for a certain fraction of time in one cannonical form and for other fractions of time in other cannonical forms. <br>(ii) The cannonical forms have no real existence. <br>(iii) There is no such equilibrium between the cannonical forms.<br>
Question 13 :
The bond dissociation energy of {tex} \mathrm { B } - \mathrm { F } {/tex} in {tex} \mathrm { BF } _ { 3 } {/tex} is {tex} 646 \mathrm { kJ } \mathrm {\ mol } ^ { - 1 } {/tex} whereas that of {tex} \mathrm { C } - \mathrm { F } {/tex} in {tex} \mathrm { CF } _ { 4 } {/tex} is {tex} 515 \mathrm { kJ } \mathrm { \ mol } ^ { - 1 } . {/tex} The correct reason for higher {tex} \mathrm { B } - \mathrm { F } {/tex} bond dissociation energy as compared to that of {tex} \mathrm { C } - \mathrm { F } {/tex} is
Question 15 :
Solubility of $NaCl$, $Na_2SO_4$ and $Na_3PO_4$ in water in increasing order is :
Question 16 :
Which of the following statements is correct in the context of the allene molecule, {tex} \mathrm {C_3H_4} {/tex}?
Question 18 :
The ratio of number of lone pairs in $NH_3$, $H_2O$ and $XeF_{2}$ is :
Question 19 :
{tex} _ { 1 } \mathrm { H } ^ { 2 } + _ { 1 } \mathrm { H } ^ { 2 } \longrightarrow _ { 2 } \mathrm { He } ^ { 3 } + _ { 0 } \mathrm { n } ^ { 1 } {/tex}<br>The above nuclear reaction is called<br>
Question 21 :
Which of the following methods is used for measuring bond length?
Question 23 :
The fifth state of matter i.e. Bose-Einstein condensate was proved in:
Question 25 :
How many grams of  aluminum will be completely oxidized by $2$ moles of oxygen at STP?
Question 27 :
When two pieces of ice are pressed together they form a single lump. This happens due to __________.
Question 28 :
Lithium reacts with water to produce hydrogen gas and lithium hydroxide. What (approx.) volume of hydrogen collected over water at 22$^oC$ and 750 mm $Hg$ pressure by the reaction of 0.208 g of $Li$?<br/>$\left[ { VP }_{ { H }_{ 2 }O }\ =\ 19.8\ mm\ Hg \right]$
Question 29 :
Assertion: On increasing the temperature, the fraction of molecules possessing most probable speed increases.
Reason: On increasing the temperature, the most probable speed of a gas increases.
Question 31 :
What volume of $NO_2$ gas at $27^oC$ and $1.0$ atm pressure would be produced?<br/>
Question 32 :
A mole always represnts for gases , 22.4 litres of volume at $ 0^0 C $ and 1 atmosphere pressure which is known as standard temperature and pressure (STP).
Question 33 :
Which one of the following represents the correct arrangement in the increasing order of forces of attraction between their particles?