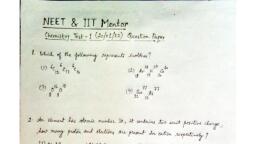Question 1 :
<span><span>Water containing more than 2% of dissolved salts and unfit for human consumption is saline water.<br/></span></span>
Question 2 :
Pollution of natural water is due to:<br/>A) Soil erosion B) Dissolved fluorides<br/>C) Soluble salts from industrial effluents<br/>
Question 3 :
______ is the contamination of water by substances harmful to life.
Question 6 :
Global warming causes:<br/>1) an increase in the sea level<br/>2) the melting of ice at mountain peaks<br/>3) a decrease in food production by plants
Question 10 :
Assertion: Chalotine sinks are formed during summer, hence, preventing ozone depletion.
Reason: In summer season, nitrogen dioxide and methane react with chlorine monoxide and chlorine radicals.
Question 12 :
Which of the following damages the ozone layer and is now the most important ozone depleting substance?
Question 13 :
The upper stratosphere consisting of the ozone layer protects us from the sun's radiation that falls in the wavelength region of:
Question 17 :
Which of the following is responsible for peeling of ozone umbrella?
Question 18 :
The gas emitted by supersonic jet planes that slowly depletes the concentration of ozone layer is
Question 20 :
Ozone in stratosphere can reduce the percentage of UV rays from sun to the extent of :<br/>
Question 22 :
{tex} \mathrm { AlI } _ { 3 } , {/tex} when reacts with {tex} \mathrm { CCl } _ { 4 } , {/tex} gives
Question 24 :
For making good quality mirrors, plates of float glass are used. These are obtained by floating molten glass over a liquid metal which does not solidify before glass. The metal used can be
Question 26 :
Which of these is not a monomer for a high molecular mass silicone polymer?
Question 28 :
Which of the following species exists (A) {tex} \left[ \mathrm { SiF } _ { 6 } \right] ^ { 2 - } , {/tex} (B) {tex} \left[ \mathrm { GeCl } _ { 6 } \right] ^ { 2 - } {/tex} and (C) {tex} \mathrm { } \left[ \mathrm { CCl } _ { 6 } \right] ^ { 2 - } ? {/tex}
Question 29 :
Bauxite ore is generally contaminated with impurity of oxides of two elements {tex}\mathrm X {/tex} and {tex}\mathrm Y . {/tex} Which of the following statement is correct?
Question 30 :
The gas evolved on heating {tex} \mathrm { CaF } _ { 2 } {/tex} and {tex} \mathrm { SiO } _ { 2 } {/tex} with concentrated {tex} \mathrm { H } _ { 2 } \mathrm { SO } _ { 4 } , {/tex} on hydrolysis gives a white gelatinous precipitate. The precipitate is:
Question 33 :
Which of the following statements are correct?<br>(i) Aluminium forms {tex} \left[ \mathrm { AlF } _ { 6 } \right] ^ { 3 - } {/tex} ion while boron forms only {tex} \left[ \mathrm { BF } _ { 4 } \right] ^ { - } {/tex} ion due to presence of {tex} d {/tex} -orbitals in aluminium.<br> (ii) The first member of a group differs from the heavier members in its ability to form p{tex} \pi {/tex} -p{tex}\pi {/tex} multiple bonds to itself and to other second row elements. While heavier member forms d{tex} \pi {/tex} -p{tex}\pi {/tex} bonds.<br>(iii) {tex} d {/tex} -orbitals contribute more to the overall stability of molecules than p{tex} \pi {/tex}-p{tex} \pi {/tex} bonds. bonding of second row elements.
Question 36 :
The correct order of increasing {tex} \mathrm { C } - \mathrm { O } {/tex} bond length of {tex} \mathrm { CO } , \mathrm { CO } _ { 2 } {/tex} and {tex} \mathrm { CO } _ { 3 } ^ { 2 - } {/tex} is:
Question 38 :
Thermal decomposition of alkanes in the absence of air is called
Question 39 :
Which of the following reactions is shown by alkynes [AMU 1984; RPMT 2000]
Question 41 :
The addition of oxygen gas to reaction mixture of chlorine and methane (photochemical chlorination):
Question 42 :
Reactivity of hydrogen atoms attached to different carbon atoms in alkanes has the order
Question 45 :
Which of the following alkane is obtained from organic matter by fermentation?<br>
Question 48 :
Which of the following is the predominant product in the reaction of <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7de02c2a2ae2953d94d2b"> with propene?
Question 50 :
Excess of {tex} \mathrm { CH } _ { 3 } \mathrm { COOH } {/tex} is reacted with {tex} \mathrm { CH } = \mathrm { CH } {/tex} in presence of {tex} \mathrm { Hg } ^ { 2 + } , {/tex} the product is
Question 52 :
A mixture of ethyl iodide and <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7dd47399925718ac6b46c"> -propyl iodide is subjected to Wurtz reaction. The hydrocarbon that will not be formed is:
Question 53 :
What happens when a mixture of acetylene and hydrogen is passed over heated Lindlar's catalyst?
Question 55 :
<img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5df322788701466b65e05029"><br>{tex} \mathrm { X } {/tex} and {tex} \mathrm { Y } {/tex} can be distinguished by<br>
Question 59 :
The total number of isotopes of hydrogen and the number of radioactive isotopes among them, respectively, are:
Question 61 :
The carbon dioxide produced in water gas shift reaction is removed by scrubbing with :
Question 62 :
In the laboratory prepration of hydrogen, pure zinc is not used because:
Question 64 :
Which of the following reactions will not produce $H_2$ gas?<br/>(a) Fe$+$dil. HCl <br/>(b) Zn$+$ dil. HCl<br/>(c) Cu$+$dil. HCl <br/>(d) Ag$+$dil. HCl <br/>
Question 67 :
Match the column - I with Column - II and pick up the correct answer:<div><br/><table class="wysiwyg-table"><tbody><tr><td>Column -I</td><td>Column -II</td></tr><tr><td>(I) Nickel</td><td>A. Conversion</td></tr><tr><td>(II) ZSM-5</td><td>B. Alkylation of benzene</td></tr><tr><td>(III) $SiO_2$</td><td>C. Hydrogenation</td></tr></tbody></table></div>
Question 68 :
In water-gas shift reaction, hydrogen gas is produced from the reaction of steam with: 
Question 69 :
Hydrogen gas is prepared in the laboratory by reacting dilute $HCl$ with granulated zinc. Following reaction takes place:<br>$Zn + 2HCl \rightarrow ZnCl_2 + H_2$<br>What would be the volume of hydrogen gas is liberated at STP when 32.65 g of zinc reacts with $HCl$?
Question 71 :
Hydrogen is evolved by the action of cold dil. $ HNO_{3} $ on