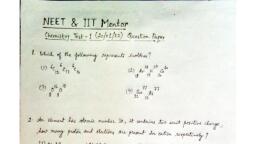Question 1 :
Henry law constant for the solubility of methane in benzene at 298K is $4.27\times { 10 }^{ 5 }$ mm Hg then the solubility of methane in benzene at 298K under 760mm Hg is:<br/>
Question 2 :
The vapour pressure of pure water at $25^{\circ}C$ is $30\ mm$. The vapour pressure of $10\%\ (W/W)$ glucose  solution at $25^{\circ}C$ is :<br/>
Question 3 :
For a given liquid at a given temperature vapour pressure is given by:<div><br/>$\;\;\;log_{10}P\:(mm)\,=\,\displaystyle-\frac{400(K)}{T}+10$</div><div><br/>Vapour pressure of the liquid at $400\:K$ is:</div>
Question 4 :
Two liquids $X$ and $Y$ form an ideal solution at $300\ K$, the vapour pressure of the solution containing $1\ mole$ of $X$ and $3\ moles$ of $Y$ is $550$ mm Hg. At the same temperature, if $1\ mole$ of $Y$ is further added to this solution, the vapour pressure of the solution increases by $10\ mm\ Hg$. Vapour pressure (in mm Hg) of $X$ and $Y$ in their pure states will be respectively :
Question 5 :
A salt which in solution gives a bluish white precipitate with $NaOH$ solution and a white precipitate with $BaCl_{2}$ solution is:
Question 6 :
The vapour pressure of two pure liquids $(A)$ and $(B)$ are $100$ and $80\ torr$ respectively. The total vapour pressure of solution obtained by mixing $2$ moles of $(A)$ and $3$ moles of $(B)$ would be :-
Question 7 :
Which one of the following statements regarding Henry's law is not correct?
Question 8 :
When a liquid that is immiscible with water was steam distilled at $95.2^o$C at a total pressure of $99.652$ kPa, the distillate contained $1.27$ g of the liquid per gram of water. What will be the molar mass of the liquid if the vapour pressure of water is $85.140$ kPa at $95.2^o$C?<br/>
Question 10 :
Choose the correct statement with respect to the vapour pressure of a liquid among the following.
Question 11 :
$H_2S$ a toxic gas with rottenegg like smell is used for the qualetative analysis, if the solubility of $H_2S$ in water,at STP is $0.195 mole.kg^{-1}$, the Henry's law constant is:
Question 12 :
To aqueous solution of Nal, increasing amounts of solid $HgI_2$ is added. The vapor pressure of the solution-
Question 13 :
Out of $Cu_2S$, $HgS$, $Ag_2S$ and $ZnS$, roasting will convert the minerals into metals in case of ___________.
Question 15 :
Extraction of gold (Au) involves the formation of complex ions X and Y.<br/>Gold ore $\displaystyle \xrightarrow [CN^-,\, H_2O,\, O_2]{Roasting}HO^-+X\xrightarrow [  ]{Zn}Y+Au $ <br/>X and Y respectively are :
Question 16 :
In the process of extraction of gold, the following reaction takes place:<div>Roasted gold ore $+ CN^- + H_2O \rightarrow [X] + OH^-$</div><div>$[X] + Zn \rightarrow [Y] + Au$ </div><div>Identify the compounds [X] and [Y].</div>
Question 17 :
Consider the following statements. Roasting is carried out to:<br/>(i) convert sulphide to oxide and sulphate.<br/>(ii) to remove impurities of $CuS$ and $FeS$ present in the ore of tin stone $(SnO_2)$ as $CuSO_4$ and $FeSO_4$.<br/>(iii) melt the ore.<br/>(iv) remove arsenic and sulphur impurities.<br/>Of these statements:<br/>
Question 18 :
Gold is also extracted by cyanide process as in case of silver.<br/><div>Write the reactions.</div>
Question 19 :
$ZnCl_{2}+NaHCO_{3}\xrightarrow [ ]{ Heat } (A)\xrightarrow [ ]{ Heat }(B)+(C)\uparrow+H_{2}O (B)+NaOH \rightarrow D$<br>Identify the compound $D$ present in the solution:
Question 21 :
Which of the following ores is not concentrated by froth floatation process?
Question 22 :
In dry cell the reaction which takes place at the zinc anode is: 
Question 23 :
Given that $E^{0}_{Cu^{2+|Cu}}=0.34\ V;E^{0}_{Ag^{+}|Ag}=0.80\ V:E^{0}_{Mg^{3+}|Mg}=-2.37$ and $E^{0} _{Al^{3+}|Al}=-1.66V$<br/>in which of the following cells the standard free energy decreases is maximum: 
Question 24 :
Consider the following standard electrode potentials $(E^o$ in volts$)$ in aqueous solution:<br>$\begin{matrix} \underline { { Element } } & \underline { { M^{ 3+ }/M } } & \underline { { M^{ + }/M } } \\ Al & -1.66 & +0.55 \\ Tl & +1.26 & -0.34 \end{matrix}$<br>Based on these data, which of the following statements is correct?
Question 25 :
Let a fully charged lead storage battery contains $1.5\ L$ of $5M\ H_2SO_4$. What will be the concentration of $H_2SO_4$ in the battery after 2.5 ampere current is drawn from the battery for 6 hour?
Question 26 :
The two aqueous solutions; $A$ $(AgNO_3)$ and $B\ (LiCl)$ were electrolysed using $Pt$ electrodes. The pH of the resulting solutions will :<br/>
Question 27 :
For a cell reaction involving two electron changes, $E_{cell}^{0} = 0.3\ V$ at $25^{\circ}C$. The equilibrium constant of the reaction is:
Question 28 :
The reduction potential of hydrogen half cell will be negative if (T=298 K) :
Question 29 :
Standard free energies of formation(in $kJ/mol$) at $298$K are $-237.2$, $-394.4$ and $-8.2$ for $H_2O(I), CO_2(g)$ and pentane(g) respectively. The value of $E^o_{cell}$ for the pentane-oxygen fuel cell is:
Question 30 :
The standard reduction potentials at 298 K for the following half-cell reactions are given:<div>$Zn^{2+} (aq) + 2e^- \rightleftharpoons Zn(s); $        $-0.762 \ V$<br/>$Cr^{3+}(aq) + 3e^- \rightleftharpoons Cr(s);$           $-0.74 \ V$<br/>$2H^+ (aq) + 2e^- \rightleftharpoons H_2(g) ;$         $+ 0.00 \ V$<br/>$Fe^{3+} (aq) + e^- \rightleftharpoons Fe^{2+} (aq) ;$   $+ 0.77 \ V$</div><div>Which one of the following is the strongest reducing agent?</div>
Question 31 :
Given that the standard reduction potentials for ${ { M }^{ + } }/{ M }$ and ${ { N }^{ + } }/{ N }$ electrodes at $298 K$ are $0.52 V$ and $0.25 V$ respectively. Which of the following is correct in respect of the following electrochemical cell?<br>$M | { M }^{ + } || { N }^{ + } | N$
Question 32 :
<b><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"></span></span></b><p class="wysiwyg-text-align-left">List I                                                   List II</p><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"></span></span><p class="wysiwyg-text-align-left">A) Salt bridge                                 1) Reduction</p><p class="wysiwyg-text-align-left">B) Standard hydrogen electrode    2) Oxidation</p><p class="wysiwyg-text-align-left">C) Cathode                                    3) Reference electrode<br/></p><p class="wysiwyg-text-align-left"><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"> D) Anode                                      4) KCl,KNO$_{3},$</span></span></p><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"></span></span><p class="wysiwyg-text-align-left"><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">                                                       (or) NH$_{4}$NO$_{3}.$</span></span><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">.</span></span></p><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"></span></span><p class="wysiwyg-text-align-left">The correct match is :</p>
Question 33 :
<div>State True or False.<br/></div>The salt bridge permits movement of charge and mixing of the solution.<br/>
Question 34 :
Ammonium dichromate is used in some fire works. The green colour powder below in air is:
Question 35 :
The atomic number of vanadium (V), chromium (Cr), manganese (Mn) and iron (Fe) are respectively 23, 24, 25 and 26 which one of these may be expected to have the highest second ionization enthalpy: 
Question 36 :
Assertion: $K_2PtCl_6$ is well-known compound whereas corresponding $Ni$ compound is not known.
Reason: $Pt^{+4}$ is thermodynamically more stable than $Ni^{+4}$.
Question 37 :
Assertion: The number of unpaired electrons in the following gaseous ions:<br>$Mn^{3+}, Cr^{3+}, V^{3+} $ and $Ti^{3+}$ are 4, 3, 2 and 1 respectively.
Reason: Among these Cr$^{3+}$ is most stable in aqueous solution.
Question 38 :
In which of the following oxoanions the oxidation state of central atom is not same as that of its group number in periodic table?
Question 39 :
Four successive members of the first row transition elements are listed below with their atomic numbers. Which one of them is expected to have the highest third ionization enthalpy? 
Question 40 :
The equivalent weight of $MnO_{2}$ is equal to its molecular weight when it is converted to:<br/>
Question 41 :
Copper becomes green when exposed to moist air for a long period. This is due to :
Question 42 :
When acetic and $K_4Fe(CN)_6$ is added to a copper salt a chocolate precipitate is obtained of the compound.<br/>
Question 43 :
Which of the following is true for the species having 3d$^4$ configuration?
Question 44 :
Which one of the following species is stable in aqueous solution<br>
Question 45 :
The equilibrium established when $HgCl_2$  is shaken with an excess of $Hg$ is: