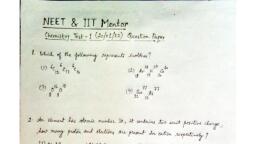Question 1 :
$0.40\ g$ of helium in a bulb at a temperature of $T\ K$ had a pressure of $p$ atm. When the bulb was immersed in water bath at temperature $50\ K$ more than the first one, $0.08\ g$ of gas had to be removed to restore the original pressure. The temperature $T$ is:
Question 2 :
When $2$ g of gas A is introduced into an evacuated flask kept at $25^oC$, the pressure was found to be $1$ atmosphere. If $3$ g of another gas B is then added to the same flask, the pressure becomes $1.5$ atm. Assuming ideal behaviour, the ratio of molecular weights $(M_B:M_A)$ is :
Question 3 :
The force which exists between particle of same kind of substance called cohesion force.
Question 4 :
When $2$ g of gas A is introduced into an evacuated flask kept at $25^oC$, the pressure was found to be $1$ atm. If $3$ g of another gas B is then added to the same flask containing A, the pressure becomes $1.5$ atm. Assuming ideal behaviour, the ratio of molecular weights $(M_A:M_B)$ is :
Question 5 :
The gaseous reaction $A_{(g)} \rightarrow 2B_{(g)} + C_{(g)}$ is found to be first order with respect to $A$. If the reaction is started with $p_{A} = 90\ mmHg$, the pressure after $10$ minutes found to be $180\ mmHg$. The rate constant of the reaction is?
Question 6 :
Which of the following statements is correct in the context of the allene molecule, {tex} \mathrm {C_3H_4} {/tex}?
Question 8 :
$KF$ combines with $HF$ to form $KHF_2$. The compound contains the species:
Question 10 :
Assertion: Sodium chloride ($NaCl$) is a stable ionic solid.
Reason: $NaCl$ has high lattice enthalpy.
Question 11 :
Molecular {tex} \mathrm { AB } {/tex} has a bond length of {tex}1.61 \ {\text{Å}}{/tex} and a dipole moment of {tex}0.38 \mathrm D{/tex}. The fractional charge on each atom (absolute magnitude) is: {tex} ( \mathrm { e } _ { 0 } = 4.802 \times 10 ^ { - 10 } \text { esu) } \quad {/tex}
Question 12 :
Dipole moment of {tex} \mathrm { H } _ { 2 } \mathrm { O } {/tex} is {tex} 1.85 \mathrm { D } {/tex}. If the bond angle is {tex} 105 ^ { \circ } {/tex} and {tex} \mathrm { O } - \mathrm { H } {/tex} bond length is {tex} 0.94 {\text{Å}}{/tex}, what is the magnitude of charge on the oxygen atom in water molecule?
Question 13 :
The bond dissociation energy of {tex} \mathrm { B } - \mathrm { F } {/tex} in {tex} \mathrm { BF } _ { 3 } {/tex} is {tex} 646 \mathrm { kJ } \mathrm {\ mol } ^ { - 1 } {/tex} whereas that of {tex} \mathrm { C } - \mathrm { F } {/tex} in {tex} \mathrm { CF } _ { 4 } {/tex} is {tex} 515 \mathrm { kJ } \mathrm { \ mol } ^ { - 1 } . {/tex} The correct reason for higher {tex} \mathrm { B } - \mathrm { F } {/tex} bond dissociation energy as compared to that of {tex} \mathrm { C } - \mathrm { F } {/tex} is
Question 14 :
Which of the following is/are misconception(s) associated with resonance?<br>(i) The molecule exists for a certain fraction of time in one cannonical form and for other fractions of time in other cannonical forms. <br>(ii) The cannonical forms have no real existence. <br>(iii) There is no such equilibrium between the cannonical forms.<br>
Question 15 :
Ionic solids do not conduct electricity, but will when the temperature is raised above the melting point. Which of the following explains why this is possible?
Question 17 :
The bond dissociation energy of {tex} \mathrm { B } - \mathrm { F } {/tex} in {tex} \mathrm { BF } _ { 1 } {/tex} is {tex} 646 \mathrm { kJ } \mathrm { mol } ^ { - 1 } {/tex} whereas that of {tex} \mathrm { C } - \mathrm { F } {/tex} in {tex} \mathrm { CF } _ { 4 } {/tex} is {tex} 515 {/tex} {tex} \mathrm { kJ } \mathrm { mol } ^ { - 1 } . {/tex} The correct reason for higher {tex} \mathrm { B } - \mathrm { F } {/tex} bond dissociation energy as compared to that of {tex} \mathrm { C } - \mathrm { F } {/tex} is
Question 18 :
$0.7\ g$ of $Na_{2}CO_{3}. xH_{2}O$ is dissolved in $100\ ml, 20\ ml$ of which required $19.8\ ml$ of $0.1\ N\ HCl$. The value of $x$ is:
Question 19 :
Assertion: Transition metals show variable valence.
Reason: Due to a large energy difference between the $ ns^2 $ and $ ( n - 1)d $ electrons.
Question 20 :
Among cellulose, polyvinyl chloride, nylon and natural rubber, in which of the polymer intermolecular force of attraction is weakest?
Question 21 :
The valency of Cr in the complex $\left[ \mathrm { Cr } \left( \mathrm { H } _ { 2 } \mathrm { O } \right) _ { 4 } \mathrm { Cl } _ { 2 } \right] ^ { + }$
Question 22 :
The longest wave length radiation emitted in the emission spectrum when the pion de-excites from n = 3 to ground state lies in which of the following region?
Question 23 :
Electrons accelerated by potential V are diffracted from a crystal. If $\mathrm{d}= 1\mathrm{A}$ and $\mathrm{i}=30^{0},\ \mathrm{V}$ should be about: <div>[$\mathrm{h}=6.6\times 10^{-34}$ Js, $\mathrm{m}_{\mathrm{e}}=9.1\times 10^{-31}$ kg, $\mathrm{e}=1.6\times 10^{-19}\mathrm{C}$]<br/></div>
Question 24 :
If the wavelength of the photon emitted from an electron jump n $=$ 4 to n $=$ 2 in a H-like species is 1216 $\overset{o}{A}$, then the species is :
Question 25 :
The energy difference (in eV) between fourth and second orbits for $H$ atom is :
Question 26 :
The wavelength($\lambda_n$) of the pion orbiting in nth stationary state is given by
Question 27 :
The only electron in the hydrogen atom resides under ordinary conditions on the first orbit. When energy is supplied, the electron moves to higher energy orbit depending on the amount of energy absorbed. When this electron returns to any of the lower orbits, it emits energy. Lyman series is formed when the electron returns the lowest orbit while Balmer series is formed when the electron returns to the second orbit. Similarly, Paschen, Brackett, and Pfind series are formed when electron returns to the third, fourth, and fifth from higher orbits, respectively.<br/>Maximum number of lines produced when an electron jumps from  nth level to ground level is equal to $\displaystyle\frac{n(n - 1)}{2}$. <br/><i></i>If the electron comes back from the energy level having energy E$_2$ to the energy level having energy E$_1$, then difference may be expressed in terms of energy of photon as<br/>E$_2$ - E$_1$ = $\Delta$E, $\lambda$ = hc/$\Delta$E<br/>Since h and c are constant, $\Delta$E corresponds to definite energy, thus, each transition from one energy level to another will produce a light of definite wavelength. This is actually observed as a line in the spectrum of hydrogen atom.<br/>Wave number of line is given by the formula<br/>v = $RZ^2\left( \displaystyle\frac{1}{n_1^2} - \frac{1}{n_2^2}\right)$<br/>where R is a Rydberg constant.The wave number of electromagnetic radiation emitted during the transition of electron in between the two levels of Li$^{2+}$ ion whose principal quantum numbers sum is 4 and difference is 2 is :
Question 28 :
If the PE of a Bohr's hydrogen atom in the ground state is zero, then its total energy in the first excited state will be :
Question 29 :
The number of revolutions of an electron in the second Bohr orbit in one second is:<br>
Question 30 :
If elements of quantum number greater than $'n'$ were not allowed, the number of possible elements in nature would have been
Question 31 :
The quantum number n of the state finally populated in $He^{+}$ ions is <br/>
Question 32 :
Assertion: 3s, 3p and 3d subshells of hydrogen have the same energy. <br/>Reason: Energy of subshells in the hydrogen atom, depends on the principal quantum number (n) and azimuthal quantum number (l). <br/>
Question 33 :
Consider a hydrogen like atom whose energy in $\displaystyle { n }^{ th }$ excited state is given by $\displaystyle { E }_{ n }=-\frac { 13.6 }{ { n }^{ 2 } } { Z }^{ 2 }$. When this excited atom makes a transition from an excited state to ground state. The most energetic photons have energy $\displaystyle { E }_{ max }=52.224eV$ and the least energetic photons have energy $\displaystyle { E }_{ min }=1.224eV$. Find the atomic number of atom.<br/>
Question 34 :
The molarity of a solution in which $5.3$g $Na_2CO_3$ is dissolved in $500$ mL will be:
Question 35 :
A $5.0$ mL solution of $H_{2}O_{2}$ liberates $0.508$ g of iodine from an acidified $KI$ solution. The strength of $H_{2}O_{2}$ solution in terms of volume strength at $STP$ will be :
Question 36 :
$300$ g of an aqueous solution of a particular solute (containing $30$% solute by mass) is mixed with $400$ g of another aqueous solution of the same solute (containing $40$% solute by mass). In the final solution, mass $\%$ of solute is :<div>[Given, Molecular mass of solute $\displaystyle = 50$]<br/></div>
Question 37 :
What $\%$ of the carbon in the $H_2CO_3-HCO^-_3$ buffer should be in the form of $HCO^-_3$ so as to have a neutral solution? $(K_a=4\times 10^{-7})$
Question 38 :
What molar concentration of $NH_3$ provides a $[OH^-]$ of $1.5 \times 10^{-3}$ ?<br>$(K_b = 1.8 \times 10^{-5})$
Question 39 :
In an ideal mixture of liquid A and B the mole fraction of A is 0.25 . If the ratio of $ P^0_A $ to $ P^0_B $ is 7/3. how many repeated distillations would be required as a minimum to obtain a small quantity of distillate which has a mole fraction of A , better than 0.80 ?
Question 40 :
If $200$ml solution of $0.5$M $NaOH$ is diluted to $800$ml then find molarity of resultant solution.
Question 41 :
Molarity of solution containing $12.6$ grams of oxalic acid in $100\ ml$ of the solution is: 
Question 42 :
Gaseous ${N}_{2}{O}_{4}$ dissociates into gaseous ${NO}_{2}$ according to the reaction ${N}_{2}{O}_{4}(g)\rightleftharpoons 2{NO}_{2}(g)$. At $300K$ and $1atm$ pressure, the degree of dissociation of ${N}_{2}{O}_{4}$ is $0.2$. If one mole of ${N}_{2}{O}_{4}$ gas is contained in a vessel, then the density of the equilibrium mixture is:
Question 43 :
$1$ mole $N_{2}$ and $4$ mole $H_{2}$ are allowed to react in a vessel and after reaction $H_{2}O$ is added to the vessel. Aqueous solution required $1$ mole $HCl$. Mole fraction of $H_{2}$ in the gaseous mixture after the reaction is:
Question 44 :
Molarity of $Ca_{3}(PO_{4})_{2}$ if the molarity of the calcium ions in the same solution is $3M$:
Question 45 :
A candle is burnt in a beaker until it extinguishes itself. A sample of gaseous mixture in the beaker contains $6.08\times 10^{20}$ molecules of $N_2, 0.76\times 10^{20}$ molecules of $O_2$, and $0.50\times 10^{20}$ molecules of $CO_2$. The total pressure is 734 mm of Hg. The partial pressure of $O_2$ would be: