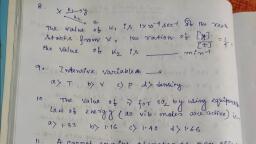Question 1 :
For the reaction <img style='object-fit:contain' src='https://storage.googleapis.com/teachmint/question_assets/JEE%20Main/5e799dfbe89a3d21421701f3' height='29' width='98' > rate is given by R = [A][B]<sup>2</sup> than the order of the reaction is
Question 2 :
Activation energy of a chemical reaction can be determined by -<br>
Question 3 :
The rate of a gaseous reaction is halved when the volume of the vessel is doubled. The order of reaction is<br>
Question 4 :
Rate of a reaction can be expressed by Arrhenius equation as <img style='object-fit:contain' src='https://storage.googleapis.com/teachmint/question_assets/JEE%20Main/5e799e0cced10523befee680' height='29' width='105' >In the equation, E represents<br>
Question 5 :
The number of molecules of the reactants taking part in a single step of the reaction tells about:
Question 6 :
The rate of the reaction, 3A + 2B → Products is given by the rate expression :Rate = k[A][B]<sup>2</sup> .If A is taken in excess, the order of the reaction would be:<br>
Question 8 :
The energies of activation for forward and reverse reactions for A2 + B<sub>2</sub><img style='object-fit:contain' src='https://storage.googleapis.com/teachmint/question_assets/JEE%20Main/5e799d58ced10523befee45a' height='18' width='39' > 2AB are 180 kJ mol<sup>-1</sup> and 200 kJ mol<sup>-1</sup> respectively. The presence of a catalyst lowers the activation energy of both (forward and reverse) reactions by 100 kJ mol<sup>-1</sup>. The enthalpy change of the reaction (A2 + B<sub>2</sub>→ 2AB) in the presence of catalyst will be (in kJ mol<sup>-1</sup>).<br>
Question 9 :
The value rate constant of a pseudo first order reaction __________.<br>
Question 10 :
For reaction A + 2B <img style='object-fit:contain' src='https://storage.googleapis.com/teachmint/question_assets/JEE%20Main/5e799d55e89a3d214216ffcc' height='21' width='36' >C + D rate law R = k[A]<sup>1</sup>[B]<sup>2</sup>. By what factor would the rate changes if concentration of A is doubled & that of B is halved ?<br>
Question 11 :
The rate constant is numerically the same for three reactions of first, second and third order respectively. Which of the following is correct : (A = reactant) -<br>
Question 12 :
The decomposition of ozone proceeds as<br/>$O_{3} \rightarrow O_{2}+O$ (fast)<br/>$O+O_{3}\rightarrow 2O_{2}$ (slow)<br/>the rate expression should be:<br/>
Question 13 :
A drop of solution (volume $0.05$ $mL$) contains $3\times { 10 }^{ -6 }$ mole of ${ H }^{ + }$. If the rate constant of disappearance of ${ H }^{ + }$ is ${ 10 }^{ 7 }$ $mol$ ${ litre }^{ -1 }$ ${ sec }^{ -1 }$, how long would it take for ${ H }^{ + }$ in the drop to disappear?
Question 14 :
Assertion:<div>Molecularity cannot be greater than three.</div><div><br/>Reason:</div><div>More than three molecules may not mutually collide with each other.<br/></div>
Question 15 :
For hypothetical chemical reaction A $\rightarrow$ I, it is found that the reaction is third order in A. What happens to the rate of reaction when the concentration of A is doubled ?
Question 16 :
A reaction follows the given concentration (M)-time graph. The rate for this reaction at 20 seconds will be :<br><img style='object-fit:contain' src='https://storage.googleapis.com/teachmint/question_assets/JEE%20Main/5e799dd0e89a3d2142170194' height='141' width='146' ><br>
Question 17 :
A hypothertical reaction, A<sub>2</sub> + B<sub>2</sub>{tex}\rightarrow{/tex}2AB follows the mechanism as given below ;<br>A<sub>2</sub><img style='object-fit:contain' src='https://storage.googleapis.com/teachmint/question_assets/JEE%20Main/5e799d8be89a3d21421700f2' height='11' width='26' >A + A ...(Fast)<br>A + B<sub>2 </sub>{tex}\rightarrow{/tex}AB + B ...(Slow)<br>A + B {tex}\rightarrow{/tex}AB ... (Fast)<br>The order of the overall reaction is -<br>
Question 18 :
The velocity constant of a reaction at $290 \,K$ was found to be $3.2 \times 10^{-3}$. At $300 \,K$ it will be:
Question 19 :
What will be the order of the reaction given below?<br/><br/><div>$NH_4CNO\rightarrow NH_2CONH_2$<br/><br/><div>The reaction is completed in three steps as:<br/><br/></div><div>(i) $NH_4CNO\rightleftharpoons NH_4NCO$ (fast equilibrium)<br/><br/></div><div>(ii) $NH_4NCO\rightleftharpoons NH_3+H-N=C=O$ (fast equilibrium)<br/><br/></div><div>(iii) $NH_3+H-N=C=O\rightarrow NH_2CONH_2$ (slow equilibrium)<br/></div></div>