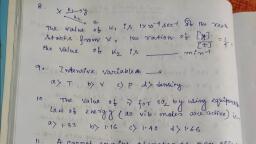Question 1 :
A following mechanism has been proposed for a reaction:<br/>$2A + B$ $\rightarrow$ $D + E$<br/>$A + B$ $\rightarrow$ $C + D$      (slow)<br/>$A + C$ $\rightarrow$ $E$     (fast)<br/>The rate law expression for the reaction is:
Question 2 :
In presence of $HCl$, sucrose gets hydrolysed into glucose and fructose. The concentration of sucrose was found to reduce from $0.4 \,M$ to $0.2 \,M$ in $1$ hour $0.1 \,M$ in $2$ hours. the order of the reaction is<br>
Question 3 :
The concentration of a reactant in a solution falls from 0.2 M to 0.1 M in 2 hours and to 0.05 M in 4 hours. The order of reaction is:
Question 4 :
In a zero order reaction, 20% of the reaction complete in 10 s. How much time it will take to complete 50% of the reaction ?
Question 5 :
For gaseous reaction, rate $k[A][B]$. If volume of container is reduced to $1/4$ of initial, then the rate of reaction will be _____ times of initial.
Question 6 :
$S_{2}O_{2} ^{2-}\rightarrow {SO_{3}}^{2-} + S$<br/>The preceding reaction was carried out in the presence of an acid-base catalyst. The reaction is found to be first order with respect to $S_{2}O_{3}^{2-}$.<br/>Which of the following statements is NOT true about the reaction?
Question 7 :
Assertion: The order of a reaction can have fractional value.
Reason: The order of a reaction cannot be written from balanced equation of a reaction.
Question 8 :
The rate of the zero order reaction; $A\, +\, B\, \rightarrow\, C$ follows the rate equation:<br/>Rate = $k[A]^0[B]^0$.<br/>Then the rate of the reaction is:<br/>
Question 9 :
The rate constant of a reaction is equal to rate of reaction:
Question 10 :
The rate constant of a first-order reaction is $3\times 10^{-6} sec^{-1}$. If the initial concentration is $0.10 \,M$, the initial rate$(in\ Ms^{-1})$ is <br>
Question 11 :
The rate of the reaction, $A+B+C\longrightarrow $ Products, is given by, $-\dfrac { d\left[ A \right]  }{ dt } =k{ \left[ A \right]  }^{ { 1 }/{ 2 } }{ \left[ B \right]  }^{ { 1 }/{ 3 } }{ \left[ C \right]  }^{ { 1 }/{ 4 } }$.<br/>The order of the reaction is:
Question 12 :
For reaction; $A\rightarrow B$, the rate of constant ${ k }_{ 1 }={ A }_{ 1 }{ e }^{ -E{ a }_{ 1 }/RT }$ and for the reaction; $P\rightarrow Q\quad $, the rate constant ${ k }_{ 2 }={ A }_{ 2 }{ e }^{ -E{ a }_{ 2 }/RT }$. If ${ A }_{ 1 }={ 10 }^{ 8 },{ A }_{ 2 }={ 10 }^{ 10 }$ and $E{ a }_{ 1 }=600,E{ a }_{ 2 }=1200$, then the temperature at which ${ k }_{ 1 }={ k }_{ 2 }$ is:
Question 13 :
For a reaction $A+B \to$ Products, the rate of the reaction was doubled when the concentration of $A$ was doubled the rate was again doubled when the conc. of $A$ & $B$ were doubled. The order of reaction with respect to $A$ & $B$ is:
Question 14 :
If the order of the reaction $x+y\xrightarrow[]{hv}xy$ is zero, it means that the rate of ____
Question 15 :
When initial concentration of a reactant is double in a reaction, its half-life period is not affected. the order of the reaction is:
Question 16 :
Which one of the following statements for order of a reaction is not correct?
Question 17 :
For the following reaction:<br>$A + 2B \rightleftharpoons C$<br>The rate law was found to be:<br>$rate = k[A]^{2}$<br>What is the overall rate order for this reaction?
Question 18 :
All of the following choices are proposed steps in the mechanism for the same overall reaction.<br>Which step would be rate determining?
Question 19 :
If the concentration of the reactants in the reaction $\:2A+B\rightarrow C+D\:$ is increased by three folds, the rate of the reaction will be increased by:
Question 20 :
Assertion: Order with respect to any reactant or product can be zero, positive, negative and fractional.
Reason: Rate cannot decrease with increase in concentration of a reactant or product.
Question 21 :
The rates of a certain reaction at different time intervals are as follows:<br/>$\begin{matrix} Time(second) & 0 & 10 & 20 \\ Rate(mol\ L^{-1}s^{-1}) & 1.8\times { 10 }^{ -2 } & 1.82\times { 10 }^{ -2 } & 1.79\times { 10 }^{ -2 } \end{matrix}$<br/>The reaction is of:
Question 22 :
The rate law of reaction between the substance $A$ and $B$ is given by $rate={K}{[A]}^{n}{[B]}^{m}$. On doubling the concentration of $A$ and making the volume of $B$ half the ratio of new rate to the earlier rate of reaction will be:
Question 23 :
Assertion: Complex reaction takes place in different step and the slowest step determines the rate of reaction.
Reason: Order and molecularity of a reaction are always equal.
Question 25 :
The decomposition of $NH_3$ on platinum surface is zero order reaction. What are the rates of production of $N_2$ and $H_2$ if $K= 2.5\times 10^{-4} mol^{-1} L s^{-1}$?
Question 26 :
Assertion: The decomposition of gaseous ammonia on a hot platinum surface is a zero order reaction at high pressure.
Reason: For a zero order reaction, the rate of reaction is independent of initial concentration.
Question 27 :
For the reaction: ${H}_{2}+{Cl}_{2}\overset { sunlight }{ \longrightarrow}2HCl$ the order of reaction is :
Question 30 :
In the case of a zero-order reaction, the ratio of time required for $75$% completion to $50$ % completion is:
Question 31 :
Which of the following is not the example of pseudounimolecular reactions?
Question 32 :
Which of the following statements are true?<div><br/>(i) Order and molecularity may be equal for some reactions.<br/>(ii) Order can be a fraction.<br/>(iii) molecularity can be a fraction.<br/>(iv) molecularity is always a whole number.<br/></div>
Question 33 :
The reaction $2X \rightarrow Y + Z$ would be zero order reaction when:
Question 34 :
Assertion: Order of a reaction with respect to any reactant can be zero, positive, negative or fractional.
Reason: Rate of a reaction cannot decrease with increase in concentration of a reactant or a product.
Question 35 :
The rate law for a reaction between the substances $A$ and $B$ is given by, $Rate=K[A]^{n}[B]^{m}$. On doubling the concentration of $A$ and halving the concentration of $B$, the ratio of the new rate to the earlier rate of the reaction will be as:
Question 36 :
In a reaction involving the synthesis of ammonia by Haber's process,<br/>$N_2+3H_2\rightleftharpoons 2NH_3,\;the\:rate\:of\:reaction\:was\:measured\:as\,=\,2.5\times 10^{-4}\:mol\:L^{-1}\,S^{-1}$.<br/>The rate of change of conc. of $H_2$ will be :
Question 38 :
If the reaction: $A + B \rightarrow C + D$ is known to be zero order, what is the expression for its rate law?
Question 39 :
For a reaction, $I^{-} + OCl^{-} \rightarrow IO^{-} + Cl^{-}$ in an aqueous medium, the rate of reaction is given by<br/><div> $\dfrac {d[IO^{-}]}{dt} = k \dfrac {[I^{-}][OCl^{-}]}{[OH^{-}]}$. The overall order of reaction is ?</div>
Question 40 :
For a certain reaction the expression for half life is  $t \propto \frac{1}{{{a^{n - 1}}}}$ then the order of reaction is: