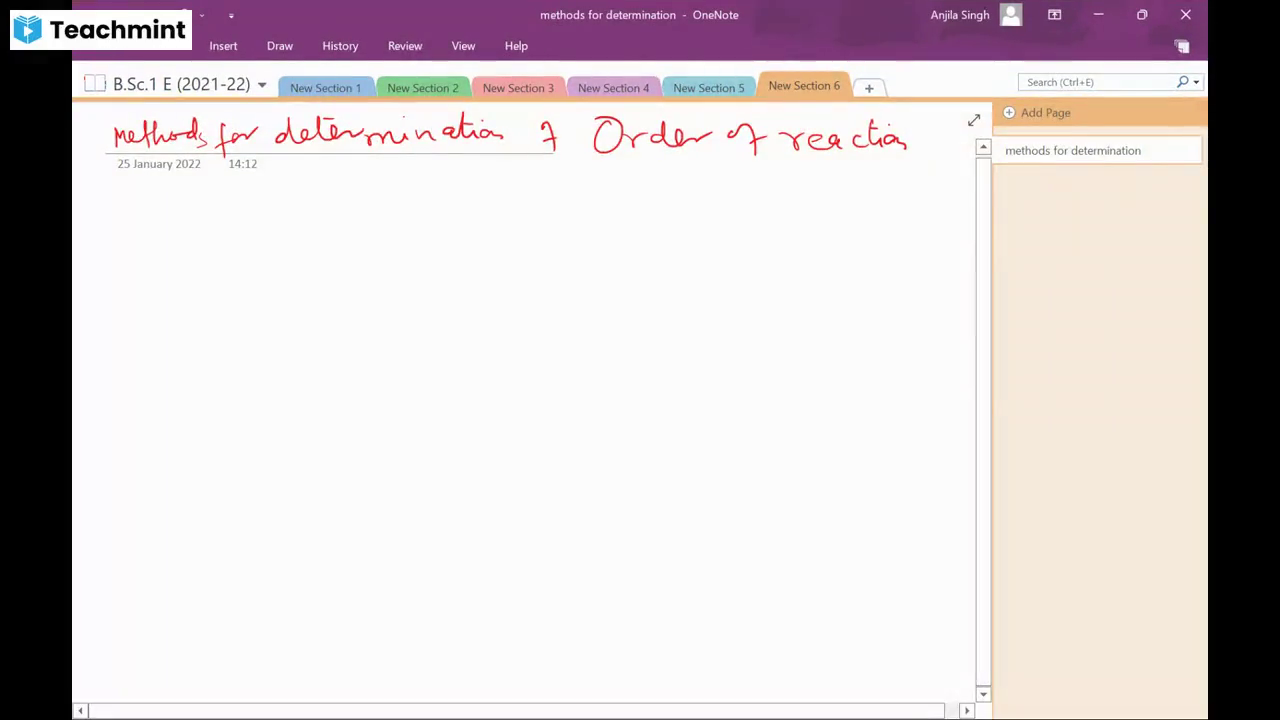
Question 1 :
In which of the compounds given below there is more than one kind of hybridization <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f625a3d2442ab8292c28"> for carbon?<br> (I) <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f625a3d2442ab8292c29"><br> <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f625a3d2442ab8292c2a">
Question 5 :
For all practical purposes, influence of inductive effect is neglected after :
Question 6 :
The sigma bond energy of <b>CH</b> bond in <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f615a3d2442ab8292bb2"> is :
Question 15 :
The number of different substitution products possible when ethane is allowed to react with bromine is sunlight are :
Question 17 :
The basicity of aniline is less than that of cyclohexylamine. This is due to
Question 18 :
The prefix name of SH group in IUPAC system is :
Question 20 :
How many chiral carbon atoms are present in 2, 3, 4- trichloropentane?
Question 23 :
29.5 mg of an organic compound containing nitrogen was digested according to Kjeldahl's method and the evolved ammonia was absorbed in 20 ml of 0.1 M HCl solution. The excess of acid required 15 ml of 0.1 M NaOH solution for complete neutralisation. The percentage of nitrogen in the compound is:
Question 25 :
Lassaigne extract is acidified with acetic acid and then lead acetate solution is added. It forms black precipitate of :
Question 26 :
Assertion: Benzene diazonium chloride does not give tests for nitrogen :
Reason: $N_{2}$ gas lose takes place during heating
Question 27 :
An organic compound weighing $0.244$ g on combustion with dry oxygen produces $0.616$ g of $CO_2$ and $0.108$ g of $H_2O$. Determine the percentage composition of the oxygen in compound.
Question 28 :
$0.186$ g of an organic compound when treated with conc. nitric acid and silver nitrate gave $0.320$ g of silver bromide. Calculate percentage of bromine in the compound.
Question 30 :
Assign number 1 for least to 4 for most to indicate the relative base strength of the following.<div><br/>$\mathop {{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}{\text{N}}{{\text{H}}_{\text{2}}}}\limits^{\text{I}} $</div><div>     <br/><span>$\mathop {{\text{p - N}}{{\text{O}}_2}{{\text{C}}_{\text{6}}}{{\text{H}}_4}{\text{N}}{{\text{H}}_{\text{2}}}}\limits^{{\text{II}}} $</span></div><div><span><span><span><br/>$\mathop {{\text{m - N}}{{\text{O}}_2}{{\text{C}}_{\text{6}}}{{\text{H}}_4}{\text{N}}{{\text{H}}_{\text{2}}}}\limits^{{\text{III}}} $</span></span></span></div><div><span><span><span>         <br/>$\mathop {{\text{m - C}}{{\text{H}}_3}{\text{O}}{{\text{C}}_{\text{6}}}{{\text{H}}_4}{\text{N}}{{\text{H}}_{\text{2}}}}\limits^{{\text{IV}}} $<br/></span></span></span></div>
Question 31 :
A Certain organic compound contains $10$% of sulphur in it. Which of the following represent the minimum molecular mass of the compound?
Question 32 :
When $FeSO_{4}$ is added in the sodium extract the compound formed is?<br/>
Question 33 :
Which reaction sequence would be best to prepare 3-chloro-aniline from benzene?
Question 35 :
In Kjeldahl's method for the estimation of nitrogen, the formula used is
Question 38 :
An organic compound which produces a bluish green coloured flame on heating in presence of copper is : <br/>
Question 39 :
If $0.765$g of an organic compound on combustion gives $0.535$g $CO_{2}$ and $0.138$g of $H_{2}O$, then the ratio of the percentage of carbon and hydrogen is:<br/>
Question 40 :
Consider the following compounds.<br><img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5df320d28701466b65e04f77"><br>The correct decreasing order of their reactivity towards hydrolysis is
Question 42 :
Blood-red colour in the detection of both N and S in Lassaigne's extract is due to the formation of__________.
Question 43 :
The order of stability of the following tautomeric compounds is:<br><img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5df320da8701466b65e04f7e"><br>
Question 44 :
<img style='object-fit:contain' src="https://data-screenshots.sgp1.digitaloceanspaces.com/5e006264ee7a9376db889ca0.jpg" /><br>The correct relation between the bond lengths {tex} a {/tex} and {tex} b {/tex} is:<br>
Question 45 :
Which of the following resonance structure is lowest in energy?<br><img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5dfc6a0cec37fa6b8da78f17"><br>
Question 47 :
The stability of carbanions in the following.<br><img style='object-fit:contain' src="https://data-screenshots.sgp1.digitaloceanspaces.com/5d6517fc96486373b3644d4f.jpg" /><br>is in the order of
Question 49 :
Assertion: Potassium can be used in Lassaigne test.
Reason: Potassium reacts vigorously.
Question 50 :
A mixture containing the following four compounds is extracted with {tex} 1 \mathrm { M \ HCl} {/tex}. The<br>compound that goes to aqueous layer is<br> <img style='object-fit:contain' src='https://storage.googleapis.com/teachmint/question_assets/NEET/5dfc6fc3ce13786b5fded60c' />