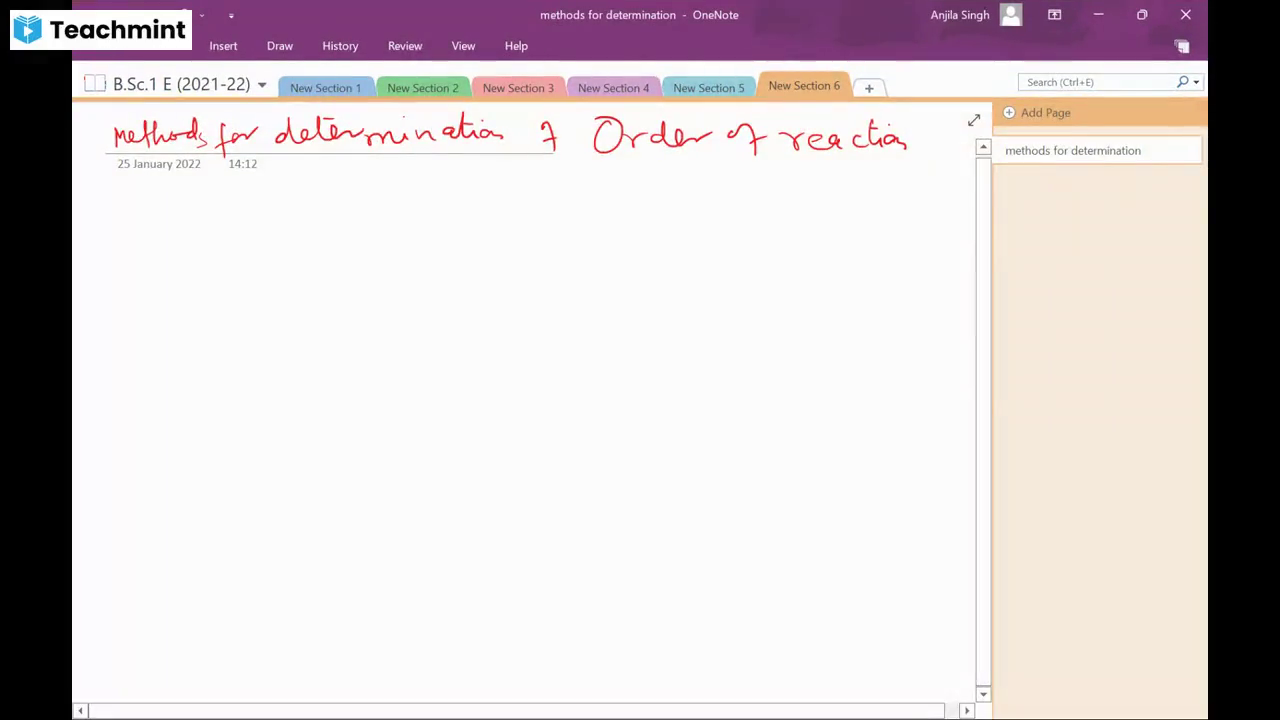
Question 1 :
How many delocalized <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5e8fdd8313cc8725d65"> -electrons are there in the compounds<br> <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5e8fdd8313cc8725d66">
Question 2 :
Crown ethers are named as <i>X</i>-crown-<i>Y</i>. In the following crown ether, <i>X </i>and <i>Y</i> are respectively<br> <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f621a3d2442ab8292c0e">
Question 3 :
The structures, <br> <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5a9fdd8313cc8725cc0"> and <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5a9fdd8313cc8725cc1"> represent:
Question 4 :
Removal of a hydride ion from a methane molecule will give a:
Question 5 :
The energy difference between the chair and boat form of cyclohexane is :
Question 6 :
Sometimes the behaviour of a compound is explained by assuming that it exists in a world between two or more different possible structures. This phenomenon is called :
Question 7 :
The function of soda lime, a mixture of solid NaOH and solid CaO during decarboxylation of carboxylic acids is :
Question 12 :
Sodium nitroprusside when added to an alkaline solution of sulphide ions produces a colouration
Question 13 :
A secondary(2 <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5c7a3d2442ab8292acd"> carbon is one that is joined to :
Question 15 :
A molecule having three different chiral carbon atoms, how many stereoisomers it will have?
Question 17 :
For all practical purposes, influence of inductive effect is neglected after :
Question 19 :
Bond energy ......... with the increase in number of lone pairs on the bonded atoms.
Question 22 :
Select the organic compounds aliphatic in nature but burn with smoky flame :
Question 24 :
The <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f50fa3d2442ab8292812"> mechanism for substitution reaction by nucleophile is favoured by :
Question 25 :
0.4 g of a silver salt of a monobasic orgainc acid gave 0.26 g pure silver on ignition. the molecular weight of the acid is (atomic weight of silver=108)
Question 26 :
Which one of the following monoenes does not exhibit geometric isomerism
Question 27 :
The shape of the <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f613a3d2442ab8292ba8"> electron cloud in acetylene is
Question 32 :
Which of the following orders regarding relative stability of free radicals is correct?
Question 38 :
The total number of cyclic structural as well as stero isomers possible for a compound with the molecular formula <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f605a3d2442ab8292b54"> is
Question 42 :
Correct order of nucleophilicity for {tex} \mathrm { SN } ^ { 2 } {/tex} reaction :-
Question 43 :
Which one of the following is the stable structure of cyclohexatriene?
Question 46 :
<img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f4f1fdd8313cc8725b43"> - pentane and 2-methyl butane are a pair of
Question 47 :
The reagent showing addition on alkene against the Markownikoffs rule of :
Question 48 :
Which among the following statements is correct with respect to the optical isomers?
Question 50 :
The correct IUPAC name of the following compound is<br> <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5d5fdd8313cc8725cfc"><br> | | |<br> <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5d5fdd8313cc8725cfd">