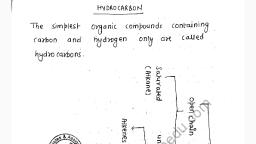
Question 4 :
Effervescence is seen when a bottle of soda water is opened.
Question 5 :
When the soil is too basic, plants do not grow well in it. To improve its quality what must be added to the soil?
Question 6 :
Assertion: pH of a neutral solution is always $7$.
Reason: pH of a solution does not depend upon temperature.
Question 9 :
If $H^{+}$ion concentration of a solution is increasedby $10$ times, its $pH$ will :<br>
Question 10 :
Aquaregia is prepared by mixing conc. $HCl$ and $conc. HNO_3$ in the ratio ________ 
Question 11 :
Which of the following methods is adopted for preparing dilute solution of ${ H }_{ 2 }S{ O }_{ 4 }$?
Question 12 :
In which of the following reactions does conc.${ H }_{ 2 }S{ O }_{ 4 }$ act as an oxidising agent?
Question 13 :
The acid present in the body of red ant is ________.<br/>
Question 16 :
When $NH_{3}(0.1\ M) 50\ ml$ mix with $HCl (0.1\ M)\ 10\ ml$ then what is $pH$ of resultant solution $(Pk_{b} = 4.75)$.
Question 19 :
Four solutions A, B, C and D when tested with universal indicator showed pH as $4, 11, 7$ and $9$ respectively. Which one of the four solutions is neutral?<br/>
Question 20 :
$1.0 \ L$ solution is prepared by mixing $61 \ gm$ benzonic acid $(pK_a = 4.2)$ with $72 \ gm$ of sodium benzoate and then $\text{300 mL 1.0 M HBr}$ solution was added. the $pH$ of final solution is:-
Question 21 :
Water of crystallization is the fixed number of water molecules chemically attached to a unit of a salt in its crystalline form.<br/>
Question 25 :
Statement I: A basic solution has more hydrogen ions than an acidic solution.<br/>Statement II: pH is defined as the -log $[H^+]$.<br/>
Question 26 :
Which colour appears when few drops of phenolphthalein put into test tube contains lime water?
Question 27 :
Which part of tooth, made up of calcium phosphate, is the hardest substance in the body?
Question 28 :
If tartaric acid is not added in baking powder, the cake will taste bitter due to te presence of -
Question 29 :
The chloride salt of a certain weak monoacidic organic base is hydrolysed to an extent of $3$% in its $0.1M$ solution at ${25}^{o}C$. Given that the ionic product of water is ${10}^{-14}$ at this temperature, what is the dissociation constant of the base?
Question 30 :
$p{ K }_{ a }$ values of four acids are given below at ${ 25 }^{ o }C$. The strongest acid is:
Question 31 :
The solution of bleaching powder in water is always a clear solution.<br/>
Question 38 :
Calculate the pH of a solution which is 0.1 M in HA and 0.5M in NaA. Ka for HA is $1.8\times { 10 }^{ -6 }$?
Question 39 :
The hydrogen ion concentration of the oceans is about $2\times10^{-9}\space M$. What is the $pH$?
Question 41 :
$pK_{a}$ of acetic acid is <b>4.741</b>. The concentration of $CH_{3}COONa$ is <b>0.01</b> M. The pH of $CH_{3}COONa$ is?
Question 44 :
A $0.01\ M$ ammonia solution is $5$% ionised, its $pH$ will be:<br/>
Question 45 :
$0.05$ mole of NaOH is added to $5$ litres of water. What will be the pH of the solution?
Question 46 :
$K_a$ for $CH_3COOH$ is $1.8\times 10^{-5}$ and $K_b$ for $NH_4OH$ is $1.8 \times 10^{-5}$. The pH of ammonium acetate will be:<br/>
Question 48 :
What is the $pH$ of a solution obtained by mixing $10$mL of $0.1$M $HCl$ and $40$ mL of $0.2$M $H_2SO_4$?
Question 50 :
The bleaching action of the bleaching powder is due to liberation of :<br/>
Question 52 :
$pH$ of saturated solution of silver salt of monobasic acid $HA$ is found ton be 9. Find the $K_{sp}$ of sparingly soluble salt $Ag A$ (s).<br/>Given : $K_a(HA)=10^{10}$
Question 53 :
1 c.c of 0.1 N $HCl$ is added to 1 litre solution of sodium chloride. The pH of the resulting solution will be:<br/>
Question 56 :
When we add a pinch of sodium hydrogen carbonate in a flask containing ethanoic acid a reaction takes place. What will be the observation when we carry out the reaction?
Question 57 :
The pair of the solutions which have pH value less than 7 is:
Question 59 :
Assertion: pH of a neutral solution is always 7
Reason: pH of a solution does not depend upon temperature
Question 60 :
When sample of baking soda is strongly ignited in crucible it suffered loss in weight of  $3.1 g $  the mass of baking soda is- 
Question 61 :
What is the $pH$ of a $0.200$ M solution of $C_6H_5COONa$? (The $K_a$ of $C_6H_5COOH$ is $6.4\times 10^{-5}$)
Question 63 :
The pH values for three different solutions are $13,\ 11$ and $9$. Arrange these pH values in the decreasing order of hydronium or ${H}^{+}$ ion concentration.
Question 65 :
Assertion: Baking powder contains $NaHCO_3$, $Ca(H_ 2PO_4)_2$, starch and $NaAl(SO_4)_2$.
Reason: $\begin{matrix}NaHCO_3\\\end{matrix} $ $Ca(H_2PO_4)_{2}$ is acidic in aqueous solution. Acidic solution of $Ca(H_2PO_4)_{2}$ liberates $CO_2$ on reaction with $NaHCO_3$Starch ] as a filter$NaAl(SO_4)^2$ retarder so that $CO_2$ is given off more slowly while preparing cake or bread.
Question 66 :
Bleaching powder is obtained by the interaction of $Cl_2$ with a:
Question 70 :
The crystalline salt, $Na_{2}SO_{4}xH_{2}O$ on heating losses $55.9\%$ of its weight. The formula of the crystalline salt is :
Question 72 :
Oxidation number of $Cl$ in ${ CaOCl }_{ 2 }$ (bleaching powder) is:
Question 73 :
State whether the given statement is true or false:<br/>Water of crystallization is the water that occurs inside crystals.
Question 75 :
The discomfort caused by indigestion due to over-eating can be cured by taking:
Question 78 :
A substances A reacts with another substances B to produce the product C and a gas D. If a mixture of the gas D and ammonia is passed through an aqueous solution of C, baking soda is formed. The substances A and B are:
Question 80 :
When bleaching powder is dissolved in water, the residue left is :<br/>
Question 81 :
The $pH$ of a $0.02\ M$ ammonia solution which is $5\%$ ionized will be:
Question 82 :
The ionization constant of a monobasic acid is $2.24\times 10^{-2}$. The $pH$ of $0.01\ M$ acid solution is:
Question 84 :
1.458 g of $Mg$ reacts with 80.0 mL of $HCl$ solution whose $pH$ is $- 0.477$. The change in $pH$ after all $Mg$ has reacted is: (Assume constant volume. $Mg = 24.3$ g $mol^{-1}$ and $log 3 = 0.477$)
Question 85 :
$ CaOCl_2 + H_2O + CO_2 \longrightarrow CaCO_3 + CaCl_2 + HOCl$ This reaction is simply:
Question 87 :
When bleaching powder undergoes auto-oxidation, the change in oxidation state is:<br/>
Question 92 :
Consider the following reactions:$CaCO_3\rightarrow CaO+CO_2$ <br> $E+CO_2\rightarrow F$ <br> $NaCl+F\rightarrow A+NaHCO_3$where $E$ and $F$ are respectively :
Question 93 :
Dissociation constants of $CH_3COOH$ and $NH_4OH$ in aqueous solution are $10^{-5}$. If pH of a $CH_3COOH$ solution is $3$. What will be the pH of $NH_4OH$?
Question 94 :
When chlorine is passed over dry slaked lime at room temperature, the main reaction product is:
Question 95 :
The more commonly used baking powder contains about 30% $NaHCO_3$, 20% $NaAl(SO_4)_2$, 10% $Ca(H_2PO_4)_2$ and 40% starch. Which of the following statements are correct?<br/>
Question 96 :
The ions responsible for the bleaching action of bleaching powder and the ions responsible for loss of bleaching action of bleaching powder due to the storage for long time respectively are:<br/>
Question 97 :
$1.0$ ml of a solution of $HCl$ with a $pH$ of $4.0$ is added it to $9.0$ ml of distilled water, the $pH$ of the final solution will be:
Question 99 :
The $pH$of $0.1 M$ solution of the following salts increases in the order
Question 101 :
The aqueous solution of which one of the following is basic?
Question 102 :
The $pH$ of a ${ 10 }^{ -9 }\ M$ solution of $HCl$ in water is :<br>
Question 103 :
$NaHCO_{3}$ can be used on its own to make cakes or bread. Baking powder is more commonly used, and contains $NaHCO_{3}$, $Ca(H_{2}PO_{4})_{2}$ and starch. An improved combination baking powder contains about 40% starch, 30% $NaHCO_{3}$ 20% $NaAl(SO_{4})_{2}$ and 10% $Ca(H_{2}PO_{4})_{2}.$ Answer the following questions based on above study.<br/>$Ca(H_{2}PO_{4})_{2}$ in baking powder:
Question 105 :
The third dissociation constant of $H_{3}PO_{4}$ is $1.3\ \times 10^{-12}$. Assume that the hydrolysis proceeds only in the first step, the $pH$ of $0.1\ M\ K_{3}PO_{4}$ solution is:
Question 106 :
Number of $OH^-$ in 1 mL solution of pH = 13 is:
Question 107 :
Iso-electric point of alanine is (pH = $6$). At which pH, maximum concentration of zwitter ion of alanine will be present?
Question 109 :
$NaHCO_{3}$ can be used on its own to make cakes or bread. Baking powder is more commonly used, and contains $NaHCO_{3}$, $Ca(H_{2}PO_{4})_{2}$ and starch. An improved combination baking powder contains about 40% starch, 30% $NaHCO_{3}$ 20% $NaAl(SO_{4})_{2}$ and 10% $Ca(H_{2}PO_{4})_{2}$Answer the following questions based on above study.<br/><br/>In improved baking powder, $NaAl(SO_{4})_{2}$ is used for :
Question 110 :
The first and second dissociation constants of an acid $H_{2}A$ are $1.0\times 10^{-5}$ and $5.0\times 10^{-10}$ respectively. The overall dissociation constant of the acid will be:
Question 111 :
To $1.0 L$ solution containing $0.1$ mol each of $NH_3$ and $NH_4Cl, 0.05$ mol NaOH is added. The change in pH will be : $(pK_a$ for $CH_3 COOH=4.74$)
Question 112 :
Percentage ionisation of water at certain temperature is $3.6\times { 10 }^{ -7 }$%. Calculate ${K}_{w}$ and $pH$ of water.
Question 113 :
When ethyl alcohol is heated with a paste of bleaching powder, we get a compound in which the function of bleaching powder is: 
Question 116 :
Which is the correct for the following reaction: <br/>$B\, (OH)_3\, +\, H_2O\, \rightarrow \, [B(OH)_4]^-\, +\, H^+$
Question 117 :
The $pH$ of a buffer solution containing equal molar concentration of a week base and its chloride ($K_{b}$ for weak base $=2\times 10^{-5}$) is :
Question 118 :
$NaHCO_{3}$ can be used on its own to make cakes or bread. Baking powder is more commonly used, and contains $NaHCO_{3}$, $Ca(H_{2}PO_{4})_{2}$ and starch. An improved combination baking powder contains about 40% starch, 30% $NaHCO_{3}$ 20% $NaAl(SO_{4})_{2}$ and 10% $Ca(H_{2}PO_{4})_{2}.$ Answer the following questions based on above study.<br/>Rise in bread and cakes is due to______ when it decomposes between $50^{o}C$ and $100^{o}C$. 
Question 119 :
The indicator constant for an acidic indicator , HIn is $ 5 \times 10^{-6}  M $. this indicator appears only in the colour of acidic from when $ \frac { [In^- ] }{ [HIn]} \le \frac {1}{20} $ and it appears only in the colour of basic form when $ \frac { [HIn]}{[In^-]} \le \frac {1}{40} . $ the pH range of indicator is:
Question 120 :
Which one of the following solutions will have the highest pH value?
Question 121 :
What is the correct increasing order of the $pH$ values of the following solutions with equal concentrations?
Question 122 :
$1\ cc$ of $0.1N$ $HCl$ is added to $1$ litre solution of sodium chloride. The $pH$ of the resulting solution will be:
Question 124 :
The $pH$ of $HCl$ is 1. $1.10\ ml$ of this solution is reacted with $40\ ml$ of $NaOH$ solution whose $pH=12$. The $pH$ of resulting solution is: $(\log(1.2)=0.108)$<br/>
Question 127 :
$\dfrac{N}{10}$ acetic acid was titrated with $\dfrac{N}{10}$ $NaOH$. when 25%, 50% and 75% of titration is over then the $pH$ of the solution will be (respectively): $[K_a = 10^5]$ 
Question 128 :
Baking powder used to make a cake is a mixture of starch, $NaHCO_3$ and $Ca(H_2PO_4)_2$. The function of $Ca(H_2PO_4)_2$ is:<br/>
Question 129 :
Substance $X$:<br/>(I) reacts with $H_2S$ to produce white turbidity.<br/>(II) changes light green solution of $FeSO_4$ into yellow colour.<br/>(III) reacts with moisture to give pungent smelling gas.<br/>Hence $X$ is:
Question 130 :
One mole of ammonia was completely absorbed in one litre solution each of<br/>I. $1\ M\ HCl$<br/>II. $1 \ M\ CH_3COOH$ <br/>III. $1\ M\ H_2SO_4$ at $298$ K<br/>The decreasing order for the $pH$ of the resulting solution is:(Given: $K_b (NH_3) = 4.74$)
Question 131 :
The $pH$ at which a 0.01 M ${ Al }^{ +3 }$ solution is 99.99% precipitated is: $\left( { K }_{ sp } \ \ Al\left( OH \right) _{ 3 }=1\times { 10 }^{ -18 }at\quad 25 \right) $
Question 132 :
The $pH$ of $0.1M$ solution of the following salts increases in the order: