Question Text
Question 1 :
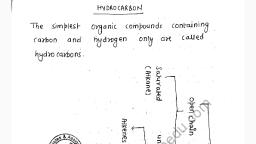
Statement I: A basic solution has more hydrogen ions than an acidic solution.<br/>Statement II: pH is defined as the -log $[H^+]$.<br/>
Question 2 :
If tartaric acid is not added in baking powder, the cake will taste bitter due to te presence of -
Question 4 :
Which part of tooth, made up of calcium phosphate, is the hardest substance in the body?
Question 7 :
The bleaching action of the bleaching powder is due to liberation of :<br/>
Question 9 :
When sample of baking soda is strongly ignited in crucible it suffered loss in weight of  $3.1 g $  the mass of baking soda is- 
Question 11 :
By which we can measure the exact $pH$ of aqueous solution?
Question 12 :
Bleaching powder is disinfectant for purification of water when water born germs are killed. But disinfectant activity is destroyed. It is due to its disproportionation into:
Question 16 :
The $pH$ at which a 0.01 M ${ Al }^{ +3 }$ solution is 99.99% precipitated is: $\left( { K }_{ sp } \ \ Al\left( OH \right) _{ 3 }=1\times { 10 }^{ -18 }at\quad 25 \right) $
Question 17 :
The indicator constant for an acidic indicator , HIn is $ 5 \times 10^{-6}  M $. this indicator appears only in the colour of acidic from when $ \frac { [In^- ] }{ [HIn]} \le \frac {1}{20} $ and it appears only in the colour of basic form when $ \frac { [HIn]}{[In^-]} \le \frac {1}{40} . $ the pH range of indicator is:
Question 18 :
The more commonly used baking powder contains about 30% $NaHCO_3$, 20% $NaAl(SO_4)_2$, 10% $Ca(H_2PO_4)_2$ and 40% starch. Which of the following statements are correct?<br/>
Question 19 :
The $pH$ of $0.1M$ solution of the following salts increases in the order:
Question 20 :
When ethyl alcohol is heated with a paste of bleaching powder, we get a compound in which the function of bleaching powder is: