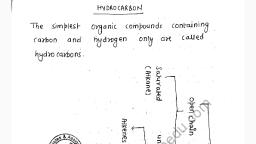
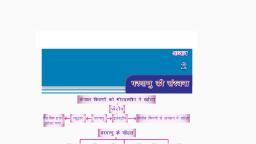
Question 2 :
When the soil is too basic, plants do not grow well in it. To improve its quality what must be added to the soil?
Question 3 :
If $H^{+}$ion concentration of a solution is increasedby $10$ times, its $pH$ will :<br>
Question 5 :
The solution of bleaching powder in water is always a clear solution.<br/>
Question 6 :
'Sodium bicarbonate solution has $pH$ less than 7.'<br/>State True or False:<br/>
Question 7 :
The bleaching action of the bleaching powder is due to liberation of :<br/>
Question 8 :
$ CaOCl_2 + H_2O + CO_2 \longrightarrow CaCO_3 + CaCl_2 + HOCl$ This reaction is simply:
Question 9 :
$H^+$ ion concentration of water does not change by adding :
Question 10 :
Bleaching powder is disinfectant for purification of water when water born germs are killed. But disinfectant activity is destroyed. It is due to its disproportionation into:
Question 11 :
Which one of the following solutions will have the highest pH value?
Question 12 :
The $pH$of $0.1 M$ solution of the following salts increases in the order
Question 13 :
The third dissociation constant of $H_{3}PO_{4}$ is $1.3\ \times 10^{-12}$. Assume that the hydrolysis proceeds only in the first step, the $pH$ of $0.1\ M\ K_{3}PO_{4}$ solution is:
Question 15 :
The ions responsible for the bleaching action of bleaching powder and the ions responsible for loss of bleaching action of bleaching powder due to the storage for long time respectively are:<br/>
Question 17 :
What is the correct increasing order of the $pH$ values of the following solutions with equal concentrations?
Question 18 :
When ethyl alcohol is heated with a paste of bleaching powder, we get a compound in which the function of bleaching powder is: 
Question 19 :
The indicator constant for an acidic indicator , HIn is $ 5 \times 10^{-6}  M $. this indicator appears only in the colour of acidic from when $ \frac { [In^- ] }{ [HIn]} \le \frac {1}{20} $ and it appears only in the colour of basic form when $ \frac { [HIn]}{[In^-]} \le \frac {1}{40} . $ the pH range of indicator is:
Question 20 :
Baking powder used to make a cake is a mixture of starch, $NaHCO_3$ and $Ca(H_2PO_4)_2$. The function of $Ca(H_2PO_4)_2$ is:<br/>