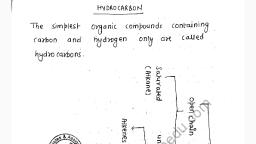
Question 1 :
When an element of very low ionization potential reacts with an element of very high electron affinity, then :
Question 5 :
The bonds present in $\left( { N }_{ 2 }{ O }_{ 5 } \right) $ are:<br/>
Question 7 :
Most of the metals which occurs in native state in nature:
Question 8 :
$2ZnS(s) + 3O_{2}(g) \xrightarrow {heat} 2ZnO(s) + 2SO_{2}(g)$ is an example of which type of reaction?
Question 10 :
Which of the following is a correct property for solid magnesium carbonate?
Question 12 :
An ore contains arsenic and antimony as impurities. How are they removed during the process of roasting?<br/>
Question 14 :
Read the following statements and answer as true or false.<br/>Metals occur in nature only as free elements.<br/>
Question 15 :
The structure of glycine (amino acid) is $H_{2}N-CH_{2}COO^{-}$ ($z$ witter ion). Select the correct statement:-
Question 16 :
Coulomb's law is used to calculate the energy of an ion pair.
Question 19 :
Assertion: Zinc is used in the galvanization of iron.
Reason: Its coating on iron articles increases their life by protecting them from rusting.
Question 23 :
Which of the following metals exist in their native state in nature?<br/>(i) Ca<br/>(ii) Au<br/>(iii) Zn<br/>(iv) Ag<br/>
Question 24 :
Separation of impurities from the extracted metals is known as __________.
Question 25 :
Which among the following metals is employed to provide cathodic protection to iron?
Question 28 :
Process of depositing a thin layer of zinc metal on iron objects is called:
Question 30 :
The process to heat the ore in the presence of excess supply of air below its melting point is called as ________.<br/>
Question 31 :
$Li$, $K$, $Ca$, $Na$, $Mg$, $Al$, $Zn$, $Fe$, $Pb$, $Cu$, $Hg$, $Ag$<br/>Which metal will displace hydrogen from acids but not water?
Question 32 :
Assertion: Ionic compounds are bad conductors of electricity.
Reason: In ionic compounds mobile ions exists.
Question 34 :
On the basis of following features identify the correct option:<br/>I. This metal is not obtained in free state.<br/>II. This metal is obtained by electrolysis of their chlorides in molten state.
Question 35 :
Galvanisation is a method of protecting iron from rusting, by coating with a thin layer of:<br/>
Question 38 :
In case of formation of ionic compounds, the electronegativity difference between them is less than 1.7.<br/>
Question 39 :
In CaF$_2$, the number of electron(s) transferred from calcium to fluorine atoms are_____.
Question 40 :
We have $CuSO_4$ solutions (blue) in two beakers A and B. In beaker A we add Ag pieces and in B we add Zn pieces. What will happen in A if :<br/>1. $Cu^{2+}$ is reduced to Cu<br/>2. no reaction.
Question 41 :
What are the conditions for a species to act as a donor and an acceptor during dative bond formation?
Question 48 :
Electrolysis is used in the extraction of metals from their ores.
Question 50 :
 Galvanizing is a process of coating iron with a metal to prevent it from rusting. The metal is :
Question 51 :
Which of the following pairs of elements form oxides of polyanions and polycations respectively:
Question 53 :
Metals react with oxygen to form their oxides which are generally
Question 54 :
Two substance $X$ and $Y$ are dissolved in water under suitable conditions. $X$ is a gas while Y is a solid under normal conditions. The solution of $Y$ is found to conduct electricity but not '$X$'. Based on the conclusion identify the nature of bond present in $X$ and $Y$ with appropriate reasons as given below. <br/>Arrange the steps given below in a sequence.<br/>(a) $Y$ is a solid at room temperature and its aqueous solution conducts electricity. This show that it is an ionic compound.<br/>(b) $X$ on dissolution in water doesn't conduct electricity and is a gas at room temperature. Hence it should be a non-polar covalent molecule.<br/>(c) Aqueous solutions of both ionic compounds and polar covalent compounds conduct electricity because of the presence of free ions.<br/>(d) All ionic compounds are solids and most of the non-polar covalent molecules are gases or solids at room temperature. Polar covalent compounds are liquids or gases.<br/>
Question 55 :
Identify the metal that occurs in the native state in nature.
Question 56 :
Identify the compound that exhibits maximum ionic character among the following:
Question 57 :
Copper occurs as a/an_________.<br/>(A) oxide ore (B) halide ore (C) carbonate ore (D) sulphide ore
Question 58 :
Which of the following combinations of combining atoms of elements gives rise to the strongest ionic bond?
Question 60 :
Reactin of NaCI with conc. $H_2SO_4$ liberates a gas X that turns moist blue litmus paper red. When gas X is passed into a test tube containing egg shell powder suspended in water another gas; Y is generated which when passed through time water makes it milky. The gases X and Y respectively, are
Question 61 :
Assertion: Sodium displaces magnesium from magnesium chloride solution.
Reason: Magnesium displaces zinc from zinc chloride solution.
Question 65 :
Metals which loses electrons more easily than hydrogen are more reactive than $H_2$ and they can :
Question 66 :
Of the following, the metals that cannot be obtained by electrolysis of the aqueous solution of their salts are ________________.
Question 67 :
Number of electrons transferred from one atom to another during bond formation in SrS (Strontium Sulphide) is :
Question 68 :
Which of the following processes is related withthe removal of sulphur by heating in the air?
Question 69 :
Which of the following oxide(s) of iron would be obtained on prolonged reaction of iron with steam?<br/>
Question 73 :
Which of the following electrolysis process is used for purification of pb?
Question 76 :
$Cl^- - e^- \rightarrow Cl$ occurs at __________ and<br>$Cl + Cl \rightarrow Cl_2$ occurs at ___________.
Question 79 :
Transfer of electrons from one atom to other atom takes place in:<br/>
Question 83 :
Metals which occurs below copper in the reactivity series of metals are:<br/>
Question 84 :
Complete the statements given below pertaining to metallurgy by filling the blank with the correct word. "M" is a metal. <br/><br/>M is ______  electropositive than hydrogen and can replace the _______ ion in hydrochloric acid to form a salt.<br/>
Question 85 :
Ultrapure elements are obtained in which of the following method of metal refining?<br/>
Question 86 :
Oxides of the various metals are converted into metal by heating but not in :
Question 87 :
Which is the correct order of formation of $MgCl_{2}$ by Born-Haber cycle ?<br/>1. Sublimation of $Mg$ ;     2. Bond dissociation of $Cl_{2}$<br/>3. Ionisation of $Mg$     ;     4. Electron gained by $Cl$
Question 89 :
All common metal chlorides are soluble in water except those of:
Question 92 :
What will be the colour of the blue cobalt chloride paper dipped in water?
Question 93 :
Out of the following reduction process-<br/>$A: { Fe }_{ 2 }{ O }_{ 3 }+C\longrightarrow Fe + CO_2$<br/>$B: ZnO+C\longrightarrow Zn + CO_2$<br/>$C: { Sn }O_{ 2 }+C\longrightarrow Sn + CO_2$<br/>$D: PbO+C\longrightarrow Pb + CO_2$<br/>Correct processes are:
Question 94 :
The electronegativities of two elements are 0.7 and 3.0. The bond formed between them would be:
Question 95 :
Assertion: Zinc metal reacts with copper sulphate solution to form zinc sulphate sulphate and copper metal.
Reason: More reactive metal displaces less reactive metal from its compound.
Question 96 :
The state in which HCl is bad conductor of electricity is _________ but, the state in which HCl is good conductor of electricity is __________.
Question 98 :
Which of the following metalsoccur in free as well as combined state?
Question 99 :
Write true or false for the following statements :<br>Copper reacts vigorously with dilute HCl .
Question 100 :
$ZnCO_3$ is precipitated from $ZnSO_4$ solution by the addition of _________.
Question 101 :
Assertion: Statement $\displaystyle -$ 1 : The first ionisation energy of N is greater than O.
Reason: Statement $\displaystyle -$ 2 : N atom has half filled p $\displaystyle -$ orbitals.
Question 104 :
Assertion: In smelting, roasted ore is heated with powdered coke in presence of a flux.
Reason: Oxides are reduced to metals by $C$ or $CO$. Impurities are removed as slag.
Question 105 :
When metallic copper comes in contact with moisture, a green powdery / pasty coating can be seen over it. This is chemically known as
Question 106 :
Which one of the following reactions will occur on heating $AgNO_3$?
Question 109 :
Assertion: Ionic bonds are directional in nature whereas covalent bonds are non-directional.
Reason: According to orbital overlap concept, the formation of a covalent bond between two atoms results by pairing of electrons present in the valence shell having same spins.
Question 110 :
$FeS + HCl \rightarrow A + B(g)$<br/><br/>Gas $B$ is passed into aqueous solution of $C$ to form $A$. Select correct statements based on the above facts.
Question 111 :
In Down's process for the extraction of sodium, the melting point of $NaCl$ is lowered from $803^{0}C$ to $600^{0}C$.<br/><br/>Choose the correct statement(s) regarding this.<br/>a) It is done in order to avoid vapourisation of metallic sodium.<br/>b) It is done in order to avoid corrosive action of sodium and chlorine on the vessel at the high temperature.<br/>c) It is done by mixing sufficient amounts of $CaCl_2$.<br/>
Question 112 :
Select the correct reason for given statement 'Fluoride of $Al$ is ionic while chloride is covalent'.
Question 115 :
Which of the following equilibria would have the lowest value of $K_{p}$ at a common temperature?
Question 118 :
Which set of compounds in the following pair of ionic compounds has the higher lattice energy?<br/>(I) $KCl$ or $MgO$<br/>(II) $LiF$ or $LiBr$<br/>(III) $MgF_2$ or $NaCl$<br/>
Question 121 :
Roasting is carried out to :1) convert sulphide to oxide and sulphate. <br/>2) remove the water of hydration. <br/>3) melt the ore. <br/>4) remove arsenic and sulphur impurities.<br/>Choose the correct options.
Question 122 :
Which of the following family form negative ions in an ionic bond?
Question 123 :
Which of the following has a bond formed from the transfer of electrons?
Question 124 :
Which of the following statement is incorrect about the extraction of lead from galena?<br>
Question 125 :
Solubility of $NaCl$, $Na_2SO_4$ and $Na_3PO_4$ in water in increasing order is :
Question 126 :
ZnO in step (ii) can also be dissolved in NaOH forming :
Question 128 :
Which of the following is not a property of an ionic compound?
Question 129 :
In $Fe-$extraction, the roasting is adopted though the ore is no having any sulphide because:
Question 133 :
Statement 1: Powdered zinc reacts faster with acid than a larger piece of zinc.<br/>Statement 2: Powdered zinc has a greater surface area.
Question 134 :
Among the following pairs of oxides, In which pair both are reduced by carbon?
Question 135 :
Mark the correct statements:<br/><br/>(i) mercury can be refined by the process of distillation.<br/><br/>(ii) In poling, the molten impure metal is stirred with green poles of wood.<br/><br/>(iii) In electrolyte refining of metals, impure metal is made as cathode and a thin strip of pure metal is made as anode.
Question 136 :
In self reduction process, the species which bring about the reduction is :<br/>
Question 137 :
Sodium is made by the electrolysis of molten mixture of about $40\% \ NaCl$ and $60\%$ $CaCl_2$ because:
Question 138 :
<p>Some of the following metals are obtained by electrolysis of their fused salts.</p><p>$Al, Na, Cu, Ag, Mg, Ca$<br/></p><p>These metals are:</p>
Question 139 :
Which of the following sets of characteristics leads to the increase in solubility of ionic substances?
Question 142 :
Amongst $LiCl$, $ BeCl_2,\ MgCl_2 $ and $RbCl$ the compounds with greatest and least ionic character, respectively are:
Question 143 :
During the formation of $NaCl$, which of the enthalpies are negative?
Question 145 :
Which one of the following metals will not reduce $ H_2 O? $
Question 146 :
Consider the following statements. Roasting is carried out to:<br/>(i) convert sulphide to oxide and sulphate.<br/>(ii) to remove impurities of $CuS$ and $FeS$ present in the ore of tin stone $(SnO_2)$ as $CuSO_4$ and $FeSO_4$.<br/>(iii) melt the ore.<br/>(iv) remove arsenic and sulphur impurities.<br/>Of these statements:<br/>
Question 147 :
Roasting of copper pyrite ores is for which of the following purpose?
Question 148 :
For purification of alumina, the modern processes most useful when (i) The impurity present is lot of iron oxides and (ii) the impurity present is a lot of silica, are
Question 149 :
Assertion (A): In the electrolysis of aqueous NaCl,Na is preferentially discharged at the mercurycathode forming sodium amalgam.<br>Reason (R) : It is due to the fact that hydrogenhas a high over voltage at mercury cathode.<br>
Question 150 :
Which of the following process is used in the extractive metallurgy of magnesium ?