Question 1 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d12273b230584979b95.JPG' />
In the above figure, what change would you observe in tube B containing calcium hydroxide solution ?
Question 2 :
State true or false: Left ventricle pumps oxygenated blood to different body parts while right ventricle pumps deoxygenated blood to lungs.
Question 3 :
Atomic number of a element is 20. What is the name of the element?
Question 4 :
The substance that triggers the fall of mature leaves and fruits from plants is ______.
Question 5 :
A person needs a lens of power –4.5 D for correction of her vision. What kind of defect in vision is she suffering from?
Question 7 :
State true or false: According to the evolutionary theory, formation of a new species is generally due to movement of individuals from one habitat to another.
Question 8 :
State true or false: The real image formed by a concave mirror is larger than the actual object when source is placed at a distance between the focus and centre of curvature of the concave mirror
Question 9 :
A metal carbonate X on reacting with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, it gives a compound Z, used for disinfecting drinking water. What is the name of the compound X.
Question 11 :
When ethanoic acid reacts with sodium hydrogencarbonate, a salt X is formed and a gas Y is evolved. What is the name of the compound Y?
Question 12 :
Atomic number of a element is 10. What is the name of the element?
Question 13 :
A metal carbonate X on reacting with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, it gives a compound Z, used for disinfecting drinking water. What is the name of the compound Y.
Question 14 :
Name the plant hormones responsible for the falling of senescent leaves.
Question 15 :
State true or false: In India the AC changes direction after every $\frac{1}{50}$ second.
Question 16 :
A non-metal A is an important constituent of our food and forms two oxides B and C. Oxide B is toxic whereas C causes global warming. Identify A.
Question 17 :
A non-metal A is an important constituent of our food and forms two oxides B and C. Oxide B is toxic whereas C causes global warming. Identify C.
Question 18 :
State true and false: Silver chloride on exposure to sunlight for long duration turns grey due to the formation of silver by decomposition of silver chloride.
Question 19 :
State true or false: The real image formed by a concave mirror is larger than the actual object when source is at a distance less than the focal length of the concave mirror.
Question 20 :
State true or false: According to the evolutionary theory, formation of a new species is generally due to accumulation of variations over several generations.
Question 21 :
State true or false: Silver chloride on exposure to sunlight for long duration turns grey due to the sublimation of silver chloride.
Question 23 :
State true or false: Sustainable development is growth that is acceptable to all the stakeholders.
Question 24 :
A non-metal A is an important constituent of our food and forms two oxides B and C. Oxide B is toxic whereas C causes global warming. To which Group of periodic table A belongs?
Question 25 :
State true or false: The real image formed by a concave mirror is larger than the actual object when source is placed at a distance greater than the radius of curvature of the concave mirror .
Question 26 :
State true or false: Unisexual flowers possessing only stamens cannot produce fruits.
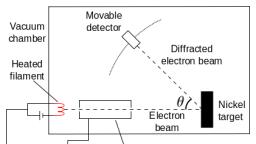
Question 27 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d0e273b230584979b90.JPG' />
In the above schematic diagram for the preparation of hydrogen gas as shown in the above figure, what would happen if same amount of zinc dust is taken in place of Zn granules in the test tube.
Question 28 :
State true or false: Left atrium receives oxygenated blood from different parts of the body while right atrium receives deoxygenated blood from lungs.
Question 30 :
State true or false: Left atrium transfers oxygenated blood to right ventricle which sends it to different parts of the body.
Question 31 :
A non-metal A is an important constituent of our food and forms two oxides B and C. Oxide B is toxic whereas C causes global warming. Identify B.
Question 32 :
State true or false: According to the evolutionary theory, formation of a new species is generally due to sudden creation by nature.
Question 33 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d14273b230584979b98.png' />
In the above given reaction, alkaline $KMnO_4$ acts as
Question 34 :
Atomic number of a element is 14. Identify the period to which this element belongs?
Question 35 :
A person needs a lens of power –4.5 D for correction of her vision. What is the focal length of the corrective lens?
Question 36 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d16273b230584979b9a.JPG' />
In the above circuits, heat produced in the resistor or combination of resistors connected to a 12 V battery will be_______.
Question 37 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d18273b230584979b9c.JPG' />
In the above figure, three 2 Ω resistors, A, B, and C, are connected. Each of them dissipates and can stand a maximum power of 18 W without melting.What is the maximum current that can flow through the B resistors.
Question 39 :
A metal carbonate X on reacting with an acid gives a gas which when passed through a solution Y gives the carbonate back. On the other hand, a gas G that is obtained at anode during electrolysis of brine is passed on dry Y, it gives a compound Z, used for disinfecting drinking water. What is the name of the compound G .
Question 40 :
State true or false: Angle of deviation is the angle , between the incident ray and the emergent ray when a light ray passes through a glass prism.
Question 41 :
Name the plant hormones responsible for the promotion of cell division.
Question 42 :
Atomic number of a element is 7. Identify the period to which this element belongs?
Question 43 :
Atomic number of a element is 7. What is the name of the element?
Question 44 :
State true or false: limitations in using solar energy can be overcome by using solar cell.
Question 46 :
A person needs a lens of power –4.5 D for correction of her vision. What is the nature of the corrective lens?
Question 47 :
State true or false: Right atrium receives deoxygenated blood from different parts of the body while left ventricle pumps oxygenated blood to different parts of the body.
Question 48 :
In an electrical circuit three incandescent bulbs A, B and C of rating 40 W, 60 W and 100 W are connected in parallel to an electric source. Which of the following is likely to happen regarding their brightness?
Question 49 :
Atomic number of a element is 14. What is the name of the element?
Question 50 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d19273b230584979b9e.JPG' />
In the above figure, three 2 Ω resistors, A, B, and C, are connected. Each of them dissipates and can stand a maximum power of 18 W without melting.What is the maximum current that can flow through the A resistors.
Question 51 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d06273b230584979b87.jpg' />
In the above image, power dissipated by 4 Ω resistor in watt is ?
Question 52 :
Three 60 W incandescent bulbs are connected in a parallel. Calculate the total power consumed.
Question 53 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d09273b230584979b8b.jpg' />
In the above image, beams of light are incident through the holes A and B and emergeout of the box through the holes C and D respectively. Which of the following could be inside the box?
Question 54 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d05273b230584979b86.jpg' />
In the above image, potential difference across 4 Ω resistor in volt is ?
Question 55 :
Identify the type of reaction: Nitrogen gas is treated with hydrogen gas in the presence of a catalyst at 773 K to form ammonia gas.
Question 57 :
State TRUE or FALSE. Solid calcium oxide reacts vigorously with water to form calcium hydroxide accompanied by liberation of heat. This process is called slaking of lime and the pH of the resulting solution will be more than seven.
Question 58 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d08273b230584979b8a.jpg' />
In the above image, the correct sequence of anaerobic reactions in yeast is ?
Question 59 :
Direct current always flows in one direction but the alternating current reverses its direction periodically. Is the statement given above about the difference between the direct and alternating current correct?
Question 60 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d04273b230584979b85.jpg' />
In the above image, current flowing through 4 Ω resistor in Ampere is ?
Question 61 :
In human beings, the statistical probability of getting either a male or female child is 50 : 50. State TRUE or FALSE.
Question 62 :
The major problem in harnessing nuclear energy is how to ?
Question 64 :
The reactivity of a piece of aluminium metal decreases if it is dipped in $HNO_3$ . State TRUE or FALSE.
Question 65 :
State TRUE or FALSE. Solid calcium oxide reacts vigorously with water to form calcium hydroxide accompanied by liberation of heat. This process is called slaking of lime and it is an endothermic reaction.
Question 66 :
State TRUE or FALSE. New species may be formed if mating does not take place.
Question 67 :
State TRUE or FALSE. Solid calcium oxide reacts vigorously with water to form calcium hydroxide accompanied by liberation of heat. This process is called slaking of lime and the pH of the resulting solution will be less than seven.
Question 68 :
State TRUE or FALSE. All green plants and blue green algae are producers
Question 69 :
'Nutrition' is a necessity for an organism. State TRUE or FALSE.
Question 70 :
Salt A commonly used in bakery products on heating converts into another salt B which is used in the manufacture of glass and a gas C is evolved. The gas C when passed through lime water, turns it milky. Identify B.
Question 71 :
State TRUE or FALSE. Solid calcium oxide reacts vigorously with water to form calcium hydroxide accompanied by liberation of heat. This process is called slaking of lime and it is an exothermic reaction.
Question 72 :
You are given the balls and sticks models of six carbon atoms and fourteen hydrogen atoms and sufficient number of sticks. In how many ways one can join the models of six carbon atoms and fourteen hydrogen atoms to form different molecules of $C_6H_{14}$ ?
Question 73 :
A magnetic compass shows a deflection when placed near a current carrying wire. The deflection of the compass get affected if the current in the wire is increased. State TRUE or FALSE.
Question 74 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d0d273b230584979b8f.jpg' />
In the above image, a constant current flows in a horizontal wire in the plane of the paper from east to west. The direction of magnetic field at a point will be North to South .............
Question 75 :
The image of a candle flame formed by a lens is obtained on a screen placed on the other side of the lens. If the image is three times the size of the flame and the distance between lens and image is 80 cm, what is the nature of the image ?
Question 76 :
Sodium carbonate is a basic salt because it is a salt of ?
Question 77 :
Identify the type of reaction Limestone is heated strongly to form quicklime and carbondioxide gas is evolved
Question 78 :
How many times does AC used in India change direction in one second?
Question 79 :
Advantage associated with water harvesting at the community level: The water can be stored during rainy season and can be used when required. State TRUE or FALSE.
Question 80 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d0a273b230584979b8c.jpg' />
In the above image, identify the circuit in which the electrical components have been properly connected.
Question 81 :
Salt A commonly used in bakery products on heating converts into another salt B which is used in the manufacture of glass and a gas C is evolved. The gas C when passed through lime water, turns it milky. Identify C.
Question 82 :
State TRUE or FALSE. Producers prepare their own food from inorganic compounds
Question 83 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d07273b230584979b88.jpg' />
In the above image, what will the difference in ammeter readings?
Question 85 :
Offspring formed as a result of sexual reproduction exhibit more variations because ?
Question 86 :
Property of the element : An element with variable (more than one) valency stored under water. Where would you locate the following element in the Periodic Table?
Question 87 :
Property of the element : A soft metal stored under kerosene. Where would you locate the following element in the Periodic Table?
Question 88 :
Sudha finds out that the sharp image of the window pane of her science laboratory is formed at a distance of 15 cm from the lens. She now tries to focus the building visible to her outside the window instead of the window pane without disturbing the lens. What is the approximate focal length of this lens ?
Question 90 :
Reproduction is essentially a phenomenon that is not for survival of an individual but for continuation of a species. State TRUE or FALSE.
Question 91 :
A student sitting at the back of the classroom cannot read clearly the letters written on the blackboard. What advice will a doctor give to her?
Question 92 :
Which one of the following four metals would be displaced from the solution of its salt by other three metals?
Question 93 :
The image of a candle flame formed by a lens is obtained on a screen placed on the other side of the lens. If the image is three times the size of the flame and the distance between lens and image is 80 cm, what is the nature of the lens ?
Question 94 :
What steps would you suggest to minimise environmental pollution caused by burning of fossil fuels?
Question 95 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d0c273b230584979b8e.jpg' />
In the above image, there are two coils wound on a non-conducting cylindrical rod. Initially the key is not inserted. Then the key is inserted and later removed. Comment about the arrangement.
Question 96 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b19d07273b230584979b89.jpg' />
In the above image, which one is the correct representation of electron dot structure of nitrogen ?
Question 98 :
In human beings, the statistical probability of getting either a male or female child is 50 : 50. The sex of an infant is determined by the type of sex chromosome contributed by the male gamete. State TRUE or FALSE.
Question 99 :
In the context of conservation of natural resources, explain the term reduce.
Question 100 :
State TRUE or FALSE. New species may be formed if chromosome number changes in the gamete