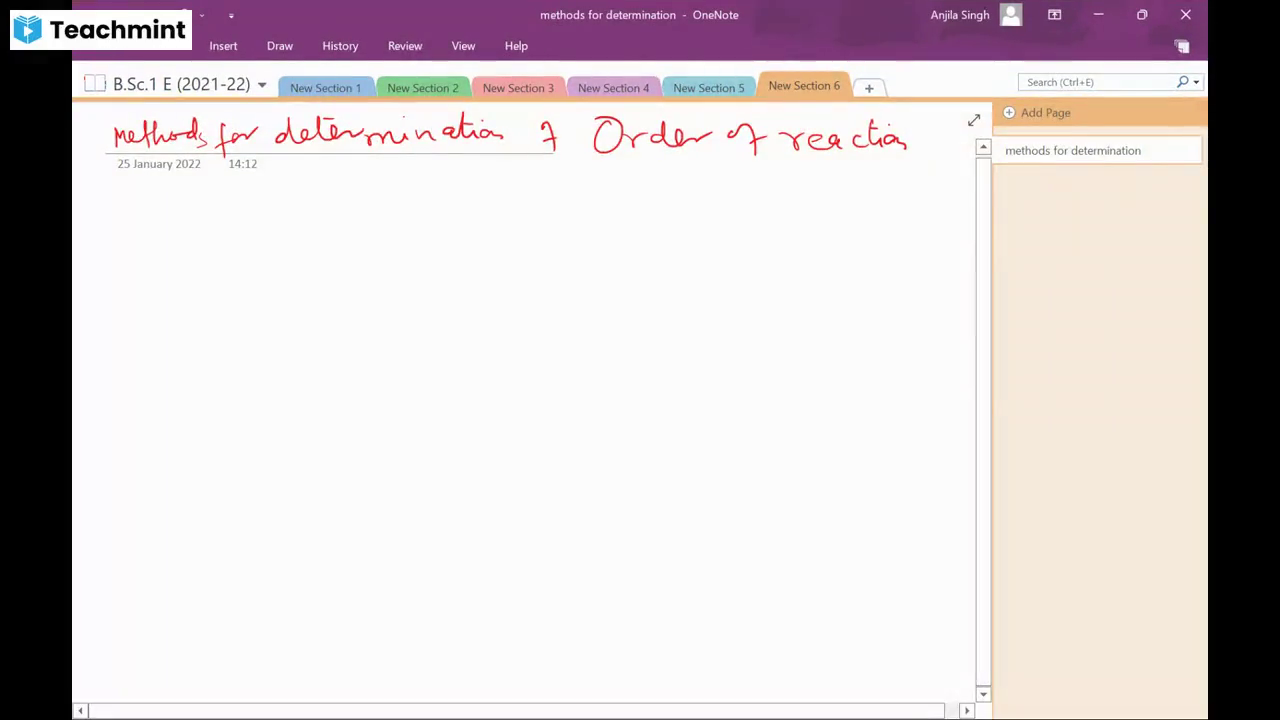
Question Text
Question 1 :
An organic compound <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f50da3d2442ab8292809"> having molecular formula <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f50ea3d2442ab829280a"> has 6 carbons in a ring system, two double bonds and also a nitro group as substitutent. The <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f50da3d2442ab8292809"> is :
Question 2 :
Which of the following sodium compound/compounds are formed when an organic compound containing both nitrogen and sulphur is fused with sodium?
Question 6 :
The production of an optically active compound from a symmetric molecule without resolution in termed as :
Question 10 :
Which one is the seniormost functional group in the nomenclature of an organic compound if it possesses more than one functional group?
Question 11 :
Mixture of sugar and common salt is separated by crystallisation by dissolving in
Question 13 :
Which type of strain is present in fully eclipsed conformation of butane?
Question 17 :
The number of different amines corresponding to the formula <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f582a3d2442ab8292a10"> is :
Question 21 :
State of hybridization of carbon atom of carbene in the singlet state is :
Question 22 :
Select the organic compound which was prepared for the first time in laboratory from its elements :
Question 23 :
The production of an optically active compound from a symmetric molecule without resolution is called :
Question 24 :
In butane, which of the following forms has the lowest energy?
Question 25 :
The maximum number of alkyl groups in <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f5b0fdd8313cc8725ce9"> is :
Question 26 :
Amongst the following which of the above are true for <img style='object-fit:contain' src="https://storage.googleapis.com/teachmint/question_assets/NEET/5ea7f58aa3d2442ab8292a41"> reaction?<br> (i) The rate of reaction is independent of the concentration of the nucleophile.<br> (ii) The nucleophile attacks the carbon atom on the side of the molecule opposite to the group being displaced.<br> (iii) the reaction proceeds with simultaneous bond formation and bond rupture.