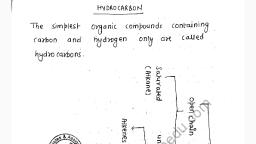
Question 2 :
The displacement of electrons in a multiple bond in the presence of attacking reagent is called:<br/>
Question 3 :
During the estimation of phosphorous present in an organic compound, which of the following reaction occurs?<br/>
Question 8 :
Compounds which have different arrangements of atoms in space while having same atoms bonded to each other are said to have:
Question 9 :
In the homologous series of alkane, the boiling point increases with :
Question 10 :
Compounds which contain oxides of carbon or carbonate hydrogencarbonate ions are called:
Question 11 :
Consider the following carbocations : <br/>(I) $ C_{6}H_{5}CH^{+}_{2} $<br/>(II) $ C_{6}H_{5}CH_{2}-CH^{+}_{2} $<br/>(III) $ C_{6}H_{5}- CH^{+} -CH_{3} $<br/>(IV) $ C_{6}H_{5}C^{+}(CH_{3})_{2} $<br/>The correct sequence for the stability of these is:
Question 14 :
A hydrocarbon has an empirical formula of $CH$ and a molar mass of $26\ g/mol$. What is the molecular formula of this compound?
Question 16 :
The correct $IUPAC$ name of the following compound $CH_{3}CH_{2}-C\equiv CH $
Question 17 :
Compounds that have the same composition but differ in structural formulas:
Question 21 :
Which of the following is the true statement for carbon atoms :<br/>
Question 24 :
You are provided with a mixture of petroleum and water. Can you separate the two by :<br/>
Question 25 :
During the estimation of phosphorous present in an organic compound, phosphorous present in the organic compound is converted into ______ .<br/>
Question 26 :
Observe the following pairs of organic compounds:<br/>(I) $C_{4}H_{9}OH$ and $C_{5}H_{11} OH$<br/>(II) $C_{7}H_{15}OH$ and $C_{5}H_{11} OH$<br/>(III) $C_{6}H_{13}OH$ and $C_{3}H_{7}OH$<br/>Which of these pairs is a homologous series according to increasing order of carbon atom?<br/>
Question 27 :
A compound of relative molecular mass 34 and empirical formula HO has the molecular formula ___________.
Question 28 :
The chemical formula which represents the actual number of atoms of each element present in a molecule is known as :
Question 33 :
The spontaneous change in specific rotation of an opacity active compound is called ?
Question 34 :
________ is process of separation whereby the components of the mixture get distributed into two liquid phases due to differences in partition coefficients during the flow of mobile phase in the chromatography column.
Question 35 :
Which of the following is <b>NOT</b> an example of an empirical formula?
Question 36 :
Which of the following forms the most stable gaseous negative ion?
Question 38 :
How many carbon atoms are present in a molecule of pentane?
Question 42 :
In which of the following pairs of resonance contributors in the structure on the left an important contributor?
Question 43 :
Which of the following is NOT an example of an molecular formula?
Question 45 :
$I$. Experimental data reveals that $1\ mole$ of a metal ($X$) combines with $3\ moles$ of a nonmetallic element ($Y$) to form a compound. The empirical formula of the compound is ${XY}_{3}$.<br/>$II$. Empirical formulas are the lowest whole number ratio of elements in a compound.
Question 48 :
A probable molecular formula for a hydrocarbon with empirical formula $CH_{3}$ is :
Question 49 :
The empirical formula shows the ......... numbers of atoms of each element in a compound. Fill in the blank.