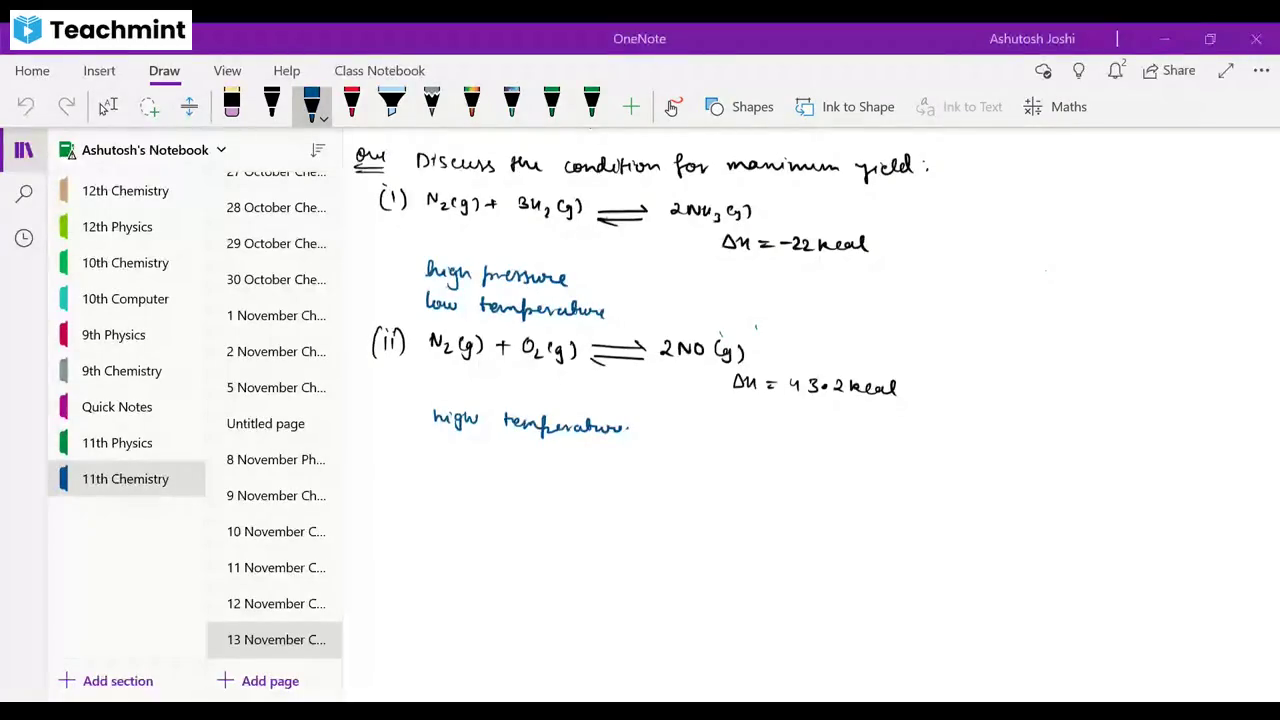
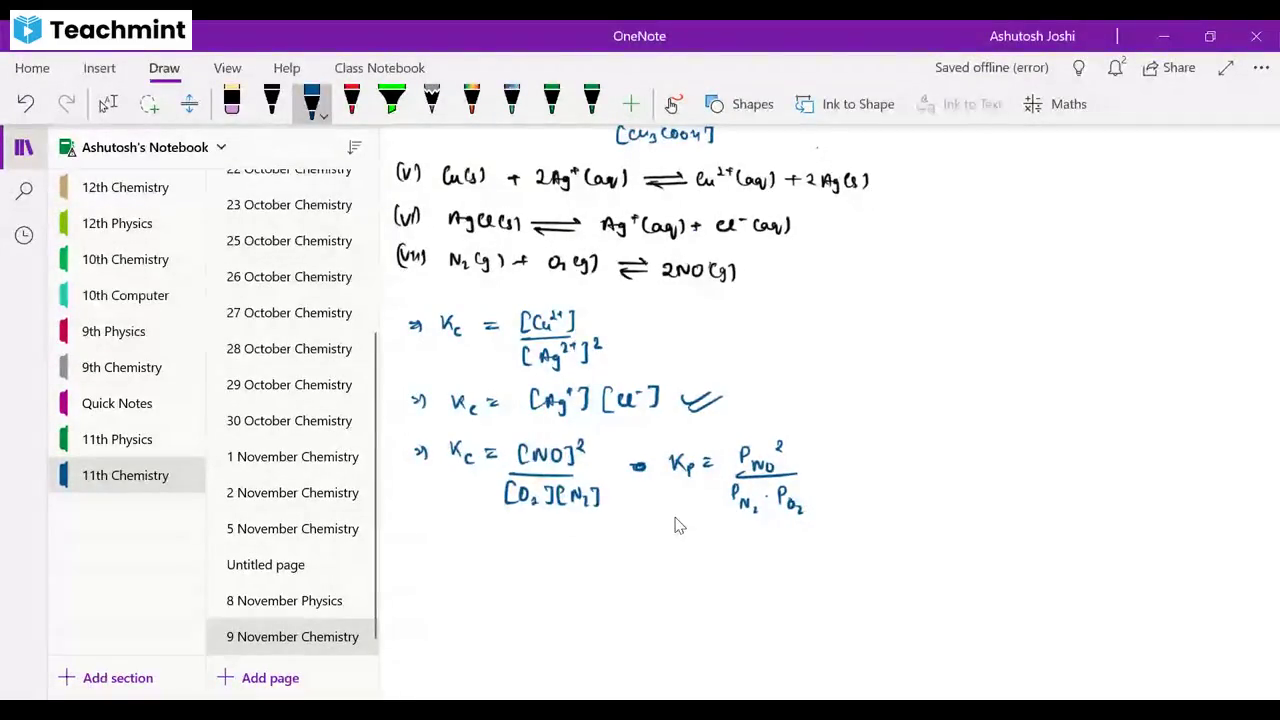
Question 1 :
An atom has electronic configuration of $2,8,18,7,$<br/>What is the atomic number the of atom?<br/>
Question 3 :
If in the ground state an atom has seven valence electrons, which of the following is the correct electronic configuration of the atom in the ground state?<br/>
Question 4 :
Which one of the following electronic configurations corresponds to the most electropositive character?
Question 5 :
With the increase in quantum number the energy difference between consecutive energy levels<br/>
Question 7 :
The valence electronic configuration of an element is $[Ne] 3s^1$. The element is :
Question 8 :
The ratio of the kinetic energy and the potential energy of electron in the hydrogen atom will be<br/>
Question 9 :
The number of unpaired electron in chromium (Atomic number = 24) is?<br/>
Question 10 :
The atomic number of an element whose differentiating electron enters in a d sub-shell:
Question 11 :
The uncertainty in the position of an electron moving with a velocity of $1 \times 10^4 \, cms^{-1}$ (accurate up to 0.011%) will be :
Question 12 :
The energy of a photon of radiation having wavelength $300 nm$ is:
Question 13 :
Imagine an atom made up of proton and a a hypothetical particle of double the mass of the electron but having the same charge as the electron. Apply the Bohr atom model and consider all possible transitions of this hypothetical particle to the first excited level. The longest wavelength photon that will be emitted has wavelength $\lambda $ (give in term of the Rydberg constant R for the hydrogen atom) equal to
Question 14 :
In which pairs do both species have the same number of unpaired electrons in p orbitals?<br>$1$ O and $Cl^+$<br>$2$. $F^+$ and $Ga^-$<br>$3$. N and $Kr^{3+}$.<br>
Question 15 :
Which one of the following sets of ions represents the collection of isoelectronic species?
Question 16 :
Among the following quantities:<br/>(i) mass number<br/>(ii) average mass of a Carbon atom in amu<br/>(iii) the charge of nucleus in amu and<br/>(iv) mass of a Carbon -12 atom in grams<br/>
Question 18 :
If the value of (n + 1) is more than 3 and less than 6, then what will be possible number of orbitals?
Question 20 :
The threshold frequency $v_0$ for a metal is $7.0\times 10^{14}sec^{-1}$. Calculate the kinetic energy of an electron emitted when radiation of frequency. $v=1.0\times 10^{15}sec^{-1}$.
Question 21 :
The radial distribution curve of the orbital with double dumbbell shape in the $4^{th}$ principal shell consists of N nodes, N is:
Question 24 :
The maximum number of electrons present in $K$ shell are _________.
Question 25 :
<table class="wysiwyg-table"><tbody><tr><td>List - I</td><td>List - II</td></tr><tr><td>A) Nodal plane</td><td>1) $\dfrac{Ze^{2}}{2r}$</td></tr><tr><td>B) p-orbital</td><td>2) $\lambda = \dfrac{h}{mv}$</td></tr><tr><td>C) de Broglie</td><td>3) Spherical</td></tr><tr><td>D) The kinetic energy of an electron</td><td>4) Probability of finding an electron is zero</td></tr><tr><td></td><td>5) Dumb bell<br/></td></tr></tbody></table><br/><br/><div>The correct match is:<br/></div>