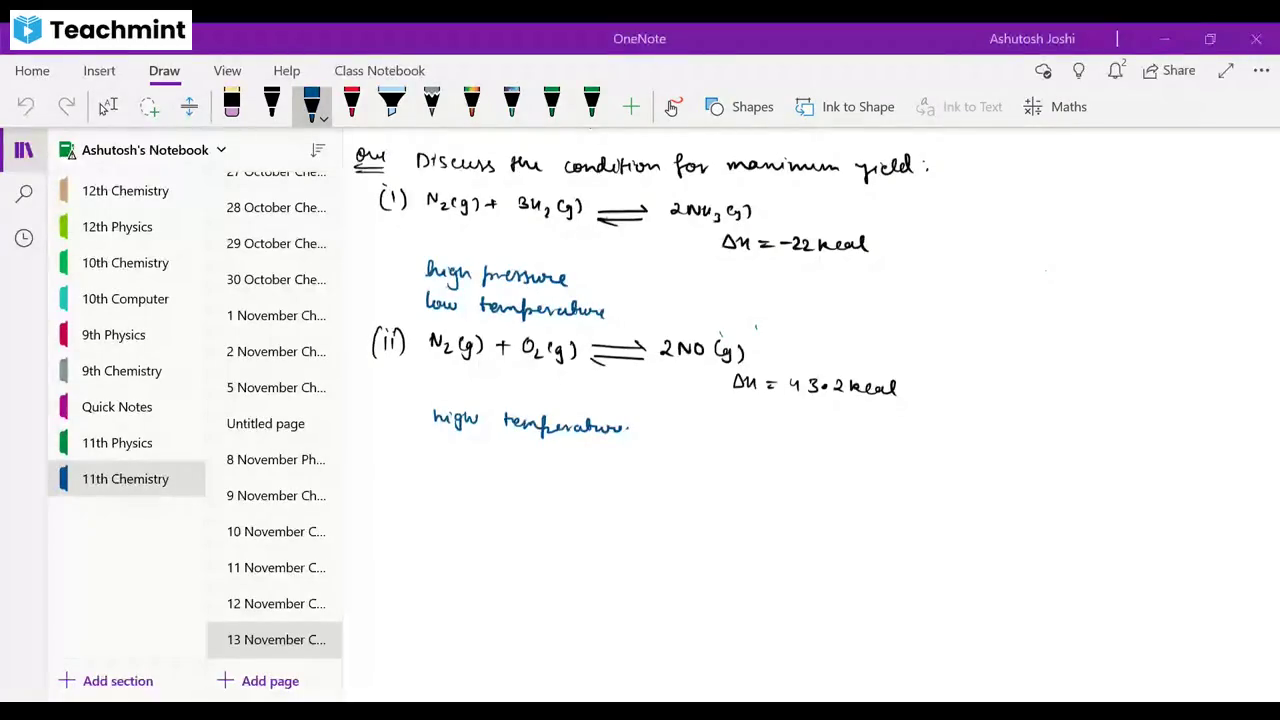
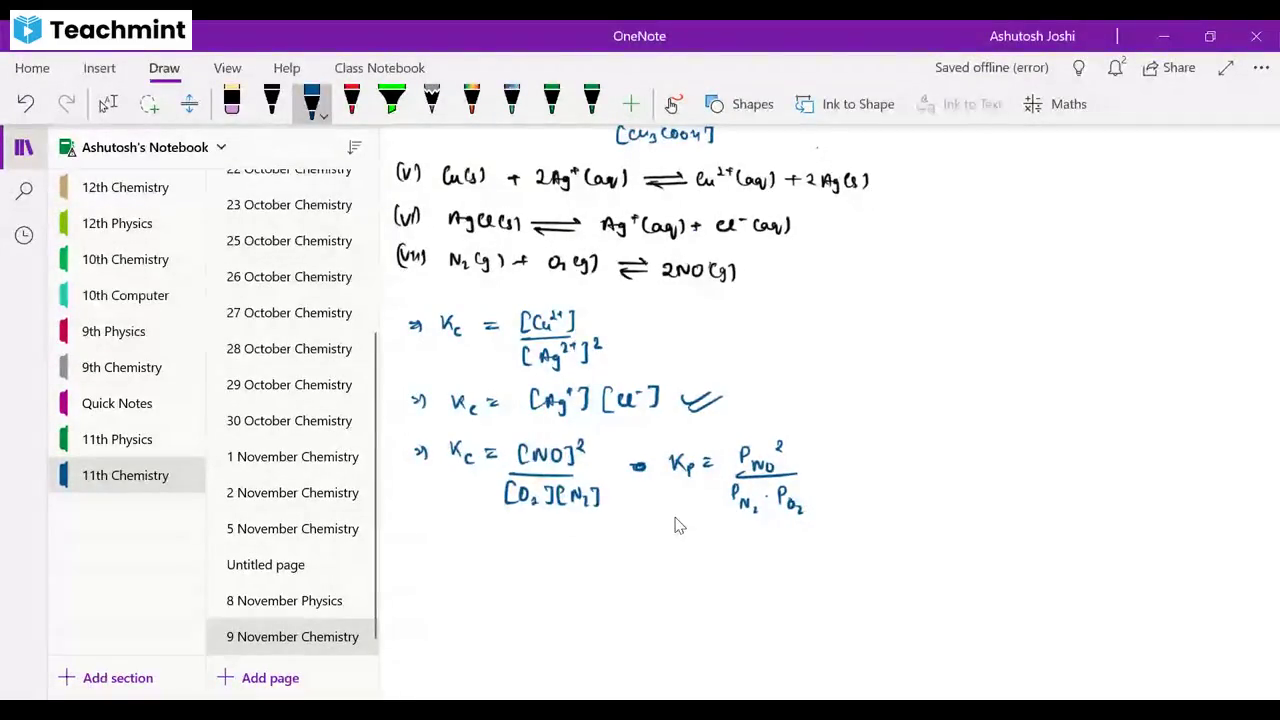
Question 1 :
An atom has electronic configuration of $2,8,18,7,$<br/>What is the atomic number the of atom?<br/>
Question 2 :
The KE of the electron in an orbit of radius 'r' in hydrogen atom is proportional to <br/>(e$=$electronic charge)<br/>
Question 3 :
The ratio of the kinetic energy and the potential energy of electron in the hydrogen atom will be<br/>
Question 4 :
The number of unpaired electron in chromium (Atomic number = 24) is?<br/>
Question 5 :
If in the ground state an atom has seven valence electrons, which of the following is the correct electronic configuration of the atom in the ground state?<br/>
Question 6 :
The electronic configuration ${ 1s }^{ 2 }{ 2s }^{ 2 }{ 2px }^{ 1 }{ 2py }^{ 1 }{ 2pz }^{ 1 }$ is of:
Question 7 :
An atom has electronic configuration 1s$^{2}$ 2s$^{2}$ 2p$^{6}$3s$^{2}$ 3p$^{6}$ 3d$^{3}$ 4s$^{2}$, you will place it in which group:
Question 8 :
If a dipositive ion has an electronic configuration of 2, 8, then the number of electrons in that ion is:
Question 9 :
The atomic number of an element whose differentiating electron enters in a d sub-shell:
Question 11 :
An isotone of Ge$_{32}^{77}$ is:<br/><br/>i. Ge$_{32}^{76}$ ii. As$_{33}^{78}$<br/><br/>iii. Se$_{34}^{79}$ iv. Se$_{34}^{78}$
Question 12 :
What is the ratio of $\Delta v= v_{max}-v_{min}$ for spectral lines corresponding to Lyman and Balmer series for hydrogen?<br/>
Question 13 :
Which one of the following set of quantum numbers is not possible for a 4p electron?
Question 14 :
The principal quantum number of an atom is related to the:
Question 19 :
The number of unpaired electrons in a chromic ion $Cr^{3+}$ $($atomic number $24)$ is:
Question 21 :
When a hydrogen atom emits a photon of energy $12.1 eV$, the orbital angular momentum changes by:
Question 22 :
An element undergoes a reaction as shown:<br>$X+2e^{-} \rightarrow X^{2-}$, energy released $=30.87 \:eV/atom$. If the energy released , is used to dissociate $4\:gms$ of $H_{2}$ molecules, equally into $H^{+}$ and $H^{*}$, where $H^{*}$ is excited state of $H$ atoms where the electron travels in orbit whose circumference equal to four times its de Broglie's wavelength. Determine the least moles of $X$ that would be required:<br>Given $I.E \: of \: H=13.6 \:eV/atom$, bond energy of $H_{2}=4.526 \:eV/molecule$
Question 23 :
When excited electrons cascade to lower energy levels in an atom then :<br/>
Question 24 :
General electronic configuration of outermost and penultimate shell is $ (n-1) s^2 (n-1)p^6 (n-1) d^x ns^2 $. If n = 4 and x = 5, then number of protons in the nucleus will be:
Question 25 :
The wave function of 2s electrons is given by : $\displaystyle \Psi _{2s}= \dfrac{1}{4\sqrt{2\pi }}\left ( \dfrac{1}{a_{0}} \right )^{3/2}\left ( 2-\dfrac{r}{a^{0}} \right )e^{\left ( 2-\frac{r}{a_{0}} \right )}$<br/>If it has a node at $r = r_{0}$, then the relation between $ r_{0}$ and $a_{0}$ is: