Question 1 :
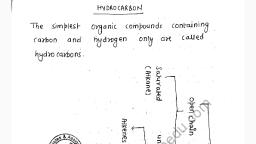
Depending on Lewis structure, $OF_2, O_2F_2$ and $S_2F_2$ which of the following statement is incorrect?
Question 2 :
A solid compound "A" consists of simple ions.<br/>Nature of the bond present in A will be:
Question 3 :
Intermolecular interactions that is dependent on the inverse cube of distance between the new molecules is
Question 5 :
In CaF$_2$, the number of electron(s) transferred from calcium to fluorine atoms are_____.
Question 7 :
Number of electrons transferred from one atom to another during bond formation in SrS (Strontium Sulphide) is :
Question 8 :
The electronegativity of Caesium is $0.7$ and that of Fluorine is $4.0$. The bond formed between the two is _________________.
Question 9 :
The electronic configuration of element $X$ is $2,8,8,1$ and the electronic configuration of element $Y$ is $2,8,7$. Then the type of bond formed between these two elements is
Question 10 :
The number of electron involved in the bond formation of $N_2$ molecule?
Question 11 :
To which of the following species, the octet rule is not applicable?
Question 14 :
Irrespective of the source, pure sample of water always yields 88.89% mass of oxygen and 11.11% mass of hydrogen. This is explained by the law of:
Question 15 :
Which set of compounds in the following pair of ionic compounds has the higher lattice energy?<br/>(I) $KCl$ or $MgO$<br/>(II) $LiF$ or $LiBr$<br/>(III) $MgF_2$ or $NaCl$<br/>
Question 16 :
The formal charges of $N_{(1)}, N_{(2)}$ and $O$ atoms in $: \overset {. .}{N_{(1)}} = N_{(2)} = \overset {. .}{O}:$ are respectively
Question 17 :
Assertion: Ionic bonds are directional in nature whereas covalent bonds are non-directional.
Reason: According to orbital overlap concept, the formation of a covalent bond between two atoms results by pairing of electrons present in the valence shell having same spins.
Question 18 :
You have three solids. One is a metal; one is an ionic compound; one is a molecular/covalent compound. You test all three solid samples to identify their bonding.<br/><table class="wysiwyg-table"><tbody><tr><td>Sample</td><td>Appearance</td><td>Melting Point $(<br/>^{\circ}C)$</td><td>Conductivity</td></tr><tr><td>A</td><td>Shiny</td><td>$962$</td><td>conductive</td></tr><tr><td>B</td><td>Crystalline</td><td>$801$</td><td>conductive in aqueous solution</td></tr><tr><td>C</td><td>Powdery</td><td>$186$</td><td>not conductive<br/></td></tr></tbody></table><br/>Based on this data, identify the type of bonds present in each sample.
Question 19 :
Which of the following species are hypervalent?<br>$1. ClO_{4}^{-}, 2. BF_{3}, 3. SO_{4}^{2-}, 4. CO_{3}^{2-}$
Question 20 :
How many dots would you put around carbon, a group IVA element?
Question 21 :
Electrons are the kind of particles which revolve around the nucleus.
Question 22 :
Goldstein's work with anode rays of $H^+$ was apparently the first observation of the :
Question 23 :
The cathode rays tube experiments carried out by J.J. Thomson demonstrated that :
Question 24 :
During the transformation of $_{ c }^{ a }{ X }$ to $_{ d }^{ b }{ Y }$, the number of $\beta $-particles emitted are
Question 27 :
The increase order for the value of $e/m$ (charge to mass ratio) for electron $(e)$, proton $(p)$; neutron $(n)$ and alpha particle $(\alpha)$ is:
Question 29 :
State True or False.<br/>High pressure and low voltage should be maintained in the discharge tube for the production of cathode rays.
Question 31 :
Assertion: e/m ratio in case of anode ray experiment is different for different gases.
Reason: The ion of gases formed after the ejection of electron are different if gas is different.
Question 33 :
If the mass of electron is doubled, the speed of electron revolving around $Li^{2+}$ nucleus will ________
Question 34 :
In an oil drop experiment, the following charges (in arbitary units) were found on a series of oil droplets:<br/>2.30 $\times$ 10$^{-15}$, 6.90 $\times$ 10$^{-15}$, 1.38 $\times$ 10$^{-14}$<br/>5.75 $\times$ 10$^{-15}$, 3.45 $\times$ 10$^{-15}$, 1.96 $\times$ 10$^{-14}$<br/>The magnitude of charge on the electron (in the same unit) is:
Question 35 :
Match the column- I with Column - II and select the correct answer:<br/><table class="wysiwyg-table"><tbody><tr><td>Column - I</td><td>Column -II</td></tr><tr><td>(I) Curie</td><td>A. $10^6 \, dis \, sec^{-1}$</td></tr><tr><td>(II) Rutherford</td><td>B. $3.7 \times 10^{10} \, dis\, sec^{-1}$</td></tr><tr><td>(III) Becquerel</td><td>C. 1 dis $sec^{-1}$</td></tr></tbody></table>
Question 36 :
Among the following, the correct statement about cathode ray discharge tube is: 
Question 37 :
The mass and charge of one mole of electrons, respectively is:
Question 39 :
Assertion: The characteristics of cathode rays do not depend upon the material of electrodes and the nature of the gas present in the cathode ray tube.
Reason: Cathode rays consist of negatively charged particles, called electrons.
Question 40 :
A beam of some kind of particle of velocity $2\times {10}^{7}m/s$ is scattered by a gold $(z=79)$ foil. Find specific charge of this particle (charge/mass) if the distance of closest approach is $7.9\times {10}^{-14}m$.
Question 41 :
The equilibrium constant for the reaction<br>$Sr(s) + Mg^{+2}(aq) \rightleftharpoons Sr^{+2}(aq) + Mg(s)$ is $4 \times 10^{12}$ at $25^o C$<br>The $E^o$ for a cell made up of the $Sr|Sr^{+2}$ and $Mg^{+2}|Mg$ half cells (log $2 = 0.3$)
Question 42 :
The following reaction that has reached equilibrium :<br/> $NaCl(s)\leftarrow \rightarrow NaCl(aq)$.<br/>What should be the nature of the solution of $NaCl$ for the phase equilibrium to exist?<br/>
Question 43 :
A chemical reaction, $A\rightleftharpoons B$, is said to be in equilibrium when:
Question 44 :
Consider the process involving gaseous species,<br/>Process I: $Cl+e^{-}\rightarrow Cl^{-}$<br/>Process II: $Na^{+}+e^{-}\rightarrow Na$<br/>Process III: $S^{-}+e^{-}\rightarrow S^{2-}$<br/>Process IV: $K\rightarrow K^{+} + e^{-}$<br/>The process in which size of product is less than reactant?<br/>
Question 45 :
A vessel contains 1 mole $PCl_{5(g)}$ at 4 atm and 0.5 mole $PCl_3$ formed at equilibrium. Now, equilibrium pressure of mixture is (assume ideal behavior)
Question 46 :
Which of the following will change the value of equilibrium constant?
Question 47 :
${X}_{2}+{Y}_{2}\rightleftharpoons 2XY$ reaction was studied at a certain temperature. In the beginning $1$ mole of ${X}_{2}$ was taken in a one litre flask and $2$ moles of ${Y}_{2}$ was taken in another $2$ litre flask. What is the equilibrium concentration of ${X}_{2}$ and ${Y}_{2}$, respectively?(Given equilibrium concentration of $[XY]=0.6{mol}^{-1}$)
Question 48 :
1 mole of '$A$', 1.5 mole of '$B$' and $2$ mole of '$C$' are taken in a vessel of volume one litre. At equilibrium concentration of '$C$' is $0.5$ mole/L. Equilibrium constant for the reaction, ${ A }_{ \left( g \right)  }+{ B }_{ \left( g \right)  }\rightleftharpoons { C }_{ \left( g \right)  }$ is:
Question 49 :
$2$ of ${ N }_{ 2 }$ is mixed with $6$ of ${ H }_{ 2 }$ in a closed vessel of one litre capacity. If 50% of ${ N }_{ 2 }$ is converted into ${ NH }_{ 3 }$ at equiibrium, the value of ${ K }_{ c }$ for the reaction ?<br/><br/>${ N }_{ 2 }\left( g \right) +3{ H }_{ 2 }\left( g \right) \rightleftharpoons 2{ NH }_{ 3 }\left( g \right) $<br/>
Question 50 :
The initial concentrations of $A$ and $B$ are same for $A = 2B \rightleftharpoons 3C$ and when equilibrium concentrations of $B$ and $C$ are same, then $K_c$ is:
Question 51 :
From application of thermodynamics on chemical reaction,<br/>we get $\Delta G = \Delta G^{0} + RT$ ln Q<br/>Also $\Delta G = \Delta H -T\Delta S$.<br/>If $\Delta G = 0$, reaction is at equilibrium. <br/>If $\Delta G > 0$ reaction is non-spontaneous under given condition. $If \Delta G < 0$, reaction is spontaneous under given condition.Consider the reaction given below.<br/>$A(s) \rightleftharpoons 2B(g) \ \ \ \ \ \ \ \ \  \Delta H^{0}= 160$ KJ/mol.<br/>$\Delta S^{0} = 400$ J/mol-K  at $400$ K<br/>Which of the following is correct at $400$ K?<br/>
Question 52 :
The equilibrium constants for the stepwise formation of $MCl$, $MCl_2$  and $MCl_3$ are $a,\ b$ and $c$ respectively. If the equilibrium constant of formation of $MCl_3$ is K, which of the following is correct?
Question 53 :
In system $A(s) \rightleftharpoons 2B(g) + 3C(g)$ at equilibrium if concentration of $'C'$ is doubled then concentration of $B$ at equilibrium is:
Question 54 :
The equilibrium constant for the isomerisation of butane at $25^oC$ is $K_c= 8$<br/>Butane $\rightarrow$ Isobutane <br/>If 5.8 g butane is introduced into a 12.5 L vessel at $25^o C$. What mass of isobutane will be present at equilibrium?
Question 55 :
$28g$ of ${N}_{2}$ and $6g$ of ${H}_{2}$ were kept at ${400}^{o}C$ in $1$ litre vessel, the equilibrium mixture contained $27.54g$ of ${NH}_{3}$. The appropriate value of ${K}_{c}$ for the above reaction can be (in ${ mole }^{ -2 }\ { litre }^{ 2 }$)
Question 56 :
Two moles of ${ N }_{ 2 }$ and two moles of ${ H }_{ 2 }$ are taken in a closed vessel of $5\ L$ capacity and suitable conditions are provided. When the equilibrium is reached, it is found that $0.6\ mole$ of ${ N }_{ 2 }$ is used up. The equilibrium concentration of ${ NH }_{ 3 }$ is________.
Question 57 :
Oxalic acid and donate two protons to water in successive reactions:<br>(1) $H_{2}C_{2}O_{4}(aq) + H_{2}O(l) \rightleftharpoons H_{3}O + (aq) + HC_{2}O_{4} - (aq)$<br>(2) $HC_{2}O_{5} - (aq) + H_{2}O(l) \rightleftharpoons H_{3}O + (aq) + C_{3}O_{4}^{2} - (aq)$<br>If $K_{cl} = 5.9\times 10^{-2}$ and $K_{c2} = 6.4\times 10^{-5}$ at $25^{\circ}C$, what is the value of $K_{c}$ for reaction $(3)$?<br>(3) $H_{2}C_{2}O_{4}(aq) + 2H_{2}O(l) \rightleftharpoons 2H_{3}O + (aq) + C_{2}O_{4}^{2} - (aq)$.
Question 58 :
$I_2+I^-\rightleftharpoons  I_3{^-}$. This reaction is set up in aqueous medium. We start with $1$ mole of $I_2$ and $0.5$ mol of $I^-$ in $1$ L flask. After equilibrium, the excess of $AgNO_3$ gave $0.25$ moles of yellow ppt. Then the equilibrium constant is: <br/>
Question 59 :
For the equilibrium $N_2+3H_2 \rightleftharpoons 2NH_3, K_c$ at 1000 $K$ is $  2.37 \times 10^{-3}$. If at equilibrium $[N_2]=2M, [H_2]=3M$, the concentration of $NH_3$ is:
Question 60 :
For the reaction ${ N }_{ 2 }(g)+3{ H }_{ 2 }(g)\rightleftharpoons 2{ NH }_{ 3 }(g)$ in a vessel, after the addition of equal number of moles of ${N}_{2}$ and ${H}_{2}$ equilibrium state is achieved. Which of the following is correct?