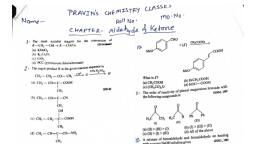
Question Text
Question 1 :
Assertion: Aldehyde have lower boiling point than ether.
Reason: Aldehydes are less polar than ether.
Question 2 :
Alcohols, aldehydes and nitriles can each be converted into carboxylic acids. Which descriptions of their conversions into carboxylic acids are correct?<table class="wysiwyg-table"><tbody><tr><td><br></td><td>alcohols</td><td>aldehydes</td><td>nitriles</td></tr><tr><td>A</td><td>hydrolysis</td><td>hydrolysis</td><td>hydrolysis</td></tr><tr><td>B</td><td>hydrolysis</td><td>hydrolysis</td><td>oxidation</td></tr><tr><td>C</td><td>oxidation</td><td>oxidation</td><td>hydrolysis</td></tr><tr><td>D</td><td>oxidation</td><td>oxidation</td><td>oxidation</td></tr></tbody></table>
Question 5 :
Structural isomerism and stereoisomerism should be considered when answering this question.<br/>The molecular formula of compound X is $C_5H_{12}O$.<br/>Compound X:<br/>$\bullet$ reacts with alkaline aqueous iodine<br/>$\bullet$ can be dehydrated to form two alkenes only.<br/>What could be the identify of compound X?<br/>
Question 6 :
Which of the following names is correct for $\underset { \overset { | }{ CHO } }{ { CH }_{ 2 } } -\underset { \overset { | }{ CHO } }{ CH } -\underset { \overset { | }{ CHO } }{ { CH }_{ 2 } }$ ?<br>
Question 7 :
Reduction of ketones cannot be carried out with which of the following reagents?<br>
Question 8 :
Ozonolysis of an organic compound gives formaldehyde as one of the products. This confirms the presence of:
Question 9 :
Action of hydrazine on aldehydes and ketones gives compound of the general structure
Question 10 :
In which of the following solvent/(s), aldehydes and ketones are soluble?<br>
Question 12 :
State whether the following statements is true or false:<br>The minimum number of carbon atoms in a ketone molecule is two.
Question 14 :
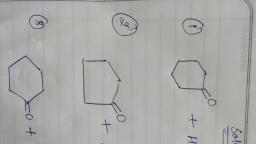
One mole of a symmetrical alkene on ozonolysis gives two moles of an aldehyde having a molecular mass of $44u$. The alkene is:
Question 15 :
$P\xrightarrow  [2.{ H }_{ 3 }{ O }^{ + } ]{ { 1.C{ H }_{ 3 }MgBr}  } R\xrightarrow [2. \Delta ]{ 1.\ dil.\ NaOH } $  4-methylpent-3-en-2-one<br/>$P$ is :