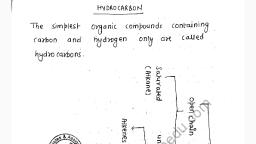
Question 4 :
What is the smallest part of the crystal that has the three-dimensional pattern of the whole lattice called?
Question 5 :
$I$. Metallic solids tend to be very brittle.$II$. Metallic bonds are broken quite easily.
Question 8 :
The nature of bonds in compounds of C and Si is:
Question 11 :
Compounds with identical crystal structure and analogous chemical formula are called:
Question 14 :
Which of the following compounds would be a white, crystalline solid?
Question 17 :
If we know the ionic radius ratio in a crystal of ionic solid, what can be known of the following?
Question 19 :
A solid melts slightly above $273$ K and is a poor conductor of heat and electricity. To which of the following categories does it belong?
Question 21 :
At very low temperature, oxygen $(O_{2})$ freezes and forms a crystalline solid. Which term best describes the solid?
Question 25 :
Which of the following is not an example of molecular crystal?<br/>
Question 26 :
Which of the following moleculeshas polar bonds but is a nonpolarmolecule?<br>
Question 28 :
Which of the following statement is not correct about molecular crystals?
Question 32 :
Which of the following exists as covalent crystals in the solid state?
Question 34 :
Assertion: $SiC$ has higher melting point that $NaCl$.
Reason: $SiC$ has stronger electronic forces of attraction.
Question 39 :
Statement: Diamond glitters because of its high refractive index.<br/><br/>State whether the given statement is true or false
Question 41 :
Examples of few solids are given below. Find out the example which is not correctly matched.
Question 44 :
Which of the following is an examples of covalent crystal solid?
Question 45 :
Which of the following exists as covalent crystals in the solid state?<br/>
Question 50 :
Statement: Covalent solids are good conductor of electricity.<br/>State whether the given statement is true or false
Question 51 :
Solid $X$ is a very hard solid which is electrical insulator in solid as well as in molten state and has extremely high melting point. What type of solid is it?
Question 58 :
The thermal conductivity of silver and copper is high because they have following binding :<br/>
Question 59 :
Which of the following is an example of metallic crystal solid?
Question 61 :
Some of the following properties are important in determining whether an element has metallic properties.<br/>I : atomic number<br/>II : atomic weight<br/>III: number of valence electrons<br/>IV: number of vacant atomic orbitals<br/>V: total number of electronic shells in the atom<br/>Select correct properties from the codes given below:
Question 62 :
Which of the solids show the following properties?<br>(i) Electrical conductivity<br>(ii) Malleability<br>(iii) Ductility<br>(iv) Fairly high melting point<br>
Question 63 :
Which type of bonding is responsible for the ability of some solid materials to conduct electricity?
Question 64 :
Statement: The melting point of all substances decreases with pressure increase.<br/><br/>State whether the given statement is true or false.
Question 68 :
Assertion: Crystalline solids can cause X-rays to diffract.
Reason: Interatomic distance in crystalline solids is of the order of $0.1$ nm.
Question 69 :
<b>Statement-I:  </b>Initially the term pseudo solid was given for solids which were easily distorted by bending and compressing forces. They even tend to flow slowly under its own weight and lose shape.<br/><b>Statement-II:</b>  These characteristics are shown by pseudo solids as in pitch, glass and thus, the name pseudo solid was replaced by supercooled liquids.<br/>
Question 70 :
The melting point ofRbBris $682^\circ C$, while that of NaFis $988^\circC$. The principal reason that melting point of $NaF$ ismuch higher than that of $RbBr$ is that:
Question 71 :
In a cubic structure of compounds which is made from $X$ and $Y$, where $X$ atoms are at the corners of the cube and $Y$ at the face centres of the cube. The molecular formula of the compound is
Question 75 :
Assertion: Crystalline solids can cause X-rays to diffract.
Reason: Interatomic distance in crystalline solids is of the order of $0.1$ nm.
Question 82 :
Assertion: Glass belongs to the category of ionic solid.
Reason: Glass is supercooled liquid.
Question 84 :
Which of the following compounds represents an inverse $2 : 3$ spinel structure?
Question 85 :
If we take 44g of${ CO }_{ 2 }$and 14g of${ N }_{ 2 }$ , what will be themole fraction of${ CO }_{ 2 }$in the mixture?
Question 87 :
In a glass we are having half filled water. Then we are continuously adding salt to the water and salt is getting dissolved into water. But a state occur when water is not able to dissolve salt. Then the solution is called as<b></b>
Question 88 :
The sum of mole fractions of solute and solvent of a solution is equal to:
Question 89 :
The mole fraction of solute in a solution is 0.3 . The mole percent of solvent is:
Question 90 :
An ideal gaseous mixture of ethane and ethene occupy $28L$ as STP. The mixture required $128g$ ${O}_{2}$ combustion, ,mole fraction of ethene in the mixture is:
Question 91 :
Which of the following are homogenous in nature ?<br>(i) Ice<br>(ii) Wood<br>(iii) Soil<br>(iv) Air
Question 92 :
One kilogram of sea water sample contains $6\ mg$ of dissolved $O_{2}$. The concentration of $O_{2}$ in the sample in ppm is:
Question 93 :
The mole fraction of solute in 10% (w/w) aqueous NaOH solution is:
Question 94 :
$Y$ molal solution of a compound in benzene has mole fraction of solute equal to $0.2$. The value of ?
Question 95 :
The sum of mole fractions of solute and solvent of a solution is equal to:
Question 96 :
The number of components in a binary solution are/is _______ .
Question 97 :
The hardness of water sample containing 0.002 mole of magnesium sulphate dissolved in a litre of water is expressed as:<br/>
Question 98 :
A mixture of ethanol and water contains $54\%$ water by mass. Calculate the mole fraction of ethanol in this solution.
Question 99 :
<p>$RH_{2}$ (ionexchange resin) can replace $Ca^{2+}$ ions in hard water as: $RH_{2}+ Ca^{2+} \rightarrowRCa +2H^{+}$.</p><p>If $1\: L$ of hard water afterpassingthrough $RH_{2}$ has $pH = 3$, then hardness in parts per million of $Ca^{2+}$is:</p>
Question 100 :
The specific gravity of a given $ H_{2}SO_{4}$ solution is $1.76$ A quantity of $3.5 ml$ of the acid is diluted to $1.0 L$ and $25 ml$ of this diluted acid required $25.6 ml$ of $N/10 (f = 095) NaOH $ solution for complete neutralization. The percentage strength (by mass) of the original acid solution is
Question 102 :
Which of the following modes of expressing concentration is/are independent of temperature?
Question 103 :
What is the degree of hardness of a sample of water containing $24\ mg$ of $MgSO_{4}$ (mol. mass $120$) per kg of water?
Question 104 :
$1$ L sample of impure water containing sulphide ion is made ammonial and is titrated with $300$ mL of $0.1\ M$ $AgNO_3$ solution. Which of the following statements is/are correct about the above reaction?
Question 105 :
One litre hard water contains $1$ mg $\displaystyle CaCl_{2}$ and $1$ mg $\displaystyle MgSO_{4}$. The hardness of water is:
Question 106 :
How much $BaCl_{2}$ would be needed to make $250\ ml$ of a solution having the same concentration of $Cl^{-}$ as one containing $3.78\, g\, NaCl$ per $100\ ml$? <br/>$\left(Ba=137\right)$<br/>
Question 107 :
The mole fraction of benzene in a solution containing $39\%$ by mass in an organic solvent of molecular mass $122$ is :
Question 108 :
Equal weights of methane and oxygen are mixed in an empty container at $25^oC$. The fraction of the total pressure exerted by oxygen is:
Question 109 :
When the solute is present in trace quantities the following expression is used:
Question 110 :
When some liquid evaporates, the average speed of the molecules remaining will .......... .
Question 111 :
If $3.65$ g of $HCl$ is dissolved in $16.2$ g of water then the mole fraction of $HCl$ ?<br/>
Question 112 :
What is the sum of the mole fractions of all the components in a three component system?
Question 115 :
A student mixes $10.0\ mL$ of $0.10\ M\ AgNO_3$ with excess copper metal. The reaction should produce $0.107$ gram of silver; however, the student obtains a mass of $150$ grams of silver. Plausible explanations for this yield $>100$% might include :<br/>I. the student did not subtract the mass of the filter paper before recording results. <br/>II. the student did not thoroughly dry the sample before massing.<br/>III. the copper metal did not react completely.<br/>
Question 116 :
$1000$ g $CaCO_3$ solution contain $10$ g carbonate. The concentration of solution as carbonate is :
Question 117 :
Assertion: The percentage weight of a compound $A$ in a solution is given by,<br> $\; \; \; \; $ % $\displaystyle of \: A = \frac{Mass \:A}{Total \: mass \: of \: solution}\times 100$<br>$Because$
Reason: The mole fraction of a component $A$ is given by,<br> $\; \; \; \; $ $\displaystyle Mole \: fraction \: of \: A =\frac{Number of \: moles \: of \: A}{ Total \: number\: of \: moles \: of \: all \: components}$
Question 118 :
The mole fraction of A in 10% (w/w) aq. solution of glucose is:
Question 119 :
The vapour pressure of a solvent decreased by $10\ mm$ of $Hg$ when a non-volatile solute was addedto the solvent. The mole fraction of solute in thesolution is $0.2$. What would be the mole fractionof solvent if decrease in vapour pressure is $20\ mm$?
Question 120 :
Two liquids A and B have vapour pressures in the ratio $P^0_A : P^0_B=2 : 3 $ at a certain temperature. Assuming that A and B form an ideal solution and ratio of mole fractions of A and B in the vapour phase is 1 : 3 , then the mole fraction of A in the solution at the same temperature is :
Question 121 :
If $5.0 \,gm$ of $BaCl_2$ is present in $10^6 \,gm$ solution, the concentration is
Question 123 :
Assertion: The sum of mole fractions of all the components of a solution is unity.
Reason: Mole fraction is a temperature dependent mode of concentration.<br/>Choose the correct option.
Question 124 :
Assertion (A): On increasing the temperature,the milli-moles of solute, milli-equivalent ofsolute,molality, mole fraction of solute and % by weightdoes not change.<br>Reason (R): Each of these involves only weightsof solute and solvent.<br>
Question 125 :
A $0.5 g$ sample of $ KH_{2}PO_{4} $ is titrated with $0.1 M$ $NaOH$. The volume of base required to do this is $25.0 ml$ The reaction is represented as :<br/>$ H_{2}PO_{4} ^{-}+OH ^{-}\rightarrow HPO_{4}^{2-}+H_{2}O $.<br/>The percentage purity of $ KH_{2}PO_{4} $ is <br/>$(K = 39, P = 31 )$
Question 126 :
On dissolving sugar in water at room temperature solution feels cool to touch. Under which of the following cases dissolution of sugar will be most rapid?
Question 127 :
1 mole of methyl alcohol is mixed with 4 moles of ethyl alcohol. The mole fraction of ethyl alcohol is:<br/>
Question 128 :
When $1.04 g$ of $BaCl_2$ is present in $105g$ of solution the concentration of solution is:
Question 129 :
A mixture has 18 g water and 414 g ethanol. The mole fraction of water in mixture is (assume ideal behaviour of the mixture) :
Question 130 :
A sample of $H_2O_2$ solution labelled as $28$ volume has density of $265$ g/L. Mark the correct option(s) representing concentration of same solution in other units.<br>
Question 131 :
The vapour pressure of a dilute aqueous solution of glucose is 750 mm of mercury at 373 K. The mole fraction of solute is:
Question 132 :
What is the equivalent mass $ IO^-_4$ when it is converted into $I_2$ in acid medium ?
Question 135 :
$14.2$ g ${Na}_{2}S{O}_{4}$ is present in ${10}^{2}$ kg water. Its concentration in ppm is:
Question 137 :
The concentration of fluoride, lead, nitrate and iron in a water sample from an underground lake was found to be $1000\ ppb, 40\ ppb, 100\ ppm$ and $0.2\ ppm$, respectively. This water is unsuitable for drinking due to high concentration of :
Question 138 :
The density of a $10.0\%$ by mass of KCl solution in water 1.06 g/mL. Calculate molarity, molality and mole fraction of KCl in this solution respectively.<br/>
Question 139 :
Assertion: A solution is a homogeneous mixture of two or more chemically non-reacting substances
Reason: Solution is prepared by the combination of two same phases of matter.
Question 141 :
Two successive reaction, $A\to B$ and $B\to C$ have yields of $90\%$ and $80\%$, respectively. What is the over all percentage yield for conversion of $A$ to $C$?
Question 142 :
$10$ g of NaCl is dissolved in $250$ g water. The correct way to express concentration of NaCl in solution are :
Question 144 :
The legal limit for human exposure to $CO$ in the work place is $35\ ppm$. Assuming that the density of air is $1.3\ g/l$, how many grams of $CO$ are in $1.0L$ of air at the maximum allowable concentration?
Question 145 :
One litre of a sample of hard water (d =$1$ g/mL) contains $136$ mg of $CaSO_{4}$ and $190$ mg of $MgCl_{2}$.What is the total hardness of water in terms of $CaCO_{3}$?
Question 146 :
$100\ ml$ of an aqueous solution contains $6.0\times {10}^{21}$ solute molecules. The solution is diluted to $1$ lit. The number of solute molecules present in $10\ ml$ of the dilute solution is:
Question 147 :
What is the percentage of silica present in the ore, by weight?
Question 149 :
The mole fraction of the solute in a $1$ molal aqueous solution is:
Question 151 :
The expression relating mole fraction of solute $(\chi_2)$ and molarity (<b>M</b>) of the solution is (where <b>d</b> is the density of the solution in $g L^{-1}$ and $Mw_1$ and $Mw_2$ are the molar masses of solvent and solute, respectively) :
Question 152 :
To determine soluble (free) $SiO_2$ in a rock, an alkaline extraction was carried out, as a result of which there was found $1.52\%$ of $SiO_2$ in the extract and also $1.02\%$ of $Al_2O_3$. Considering that, apart from the free $SiO_2$, the extract also contained the $SiO_2$, that had passed into it from Kaolin $(2SiO_2, Al_2O_3)$, the percentage of $SiO_2$, in the rock being analysed is $(Si=28, Al=27)$
Question 153 :
When $25g$ of ${Na}_{2}{SO}_{4}$ is dissolved in ${10}^{3}kg$ of solution, its concentration will be :
Question 154 :
What is the mole fraction of the solute in a $1.00$ m aqueous solution?
Question 155 :
$2.0\ g$ mixture of sodium carbonate and sodium bicarbonate on heating to constant weight gave $224\ mL$ of $CO_{2}$ at N.T.P.<br>The $\%$ weight of sodium bicarbonate in the mixture is:
Question 156 :
$10$ L of hard water required $0.56$ g of lime (CaO) for removing hardness. Hence, the temporary hardness in ppm (part per million) of $CaCO_3$ is:
Question 157 :
A mixture of $a$ mol of $C_3H_8$ and $b$ mol of $C_2H_4$ was kept is a container of $V$ L which exerts a pressure of $4.93$ atm at temperature T. Mixture was burnt in presence of $O_2$ to convert $C_3H_8$ and $C_2H_4$ into $CO_2$ in the container at the same temperature. The pressure of gases after the reaction and attaining the thermal equilibrium with atmosphere at temperature T was found to be $11.08$ atm.<br/><br/>The mole fraction of $C_3H_8$ in the mixture is :<br/>
Question 158 :
$10$ L of hard water required $0.56$ g of lime $(CaO)$ for removing hardness. Hence, temporary hardness in ppm of $CaCO_3$ is:<br>
Question 159 :
At $88^0 C$, benzene has a vapour pressure of 900 torr and toluene has a vapour pressure of 360 torr. What is the mole fraction of benzene in the mixture with toluene that will boil at $88^0 C$ at 1 atm pressure, benzene -toluene from an ideal solution?
Question 160 :
The mole fraction of water in 98% (w/w) $H_2SO_4$ solution is :
Question 161 :
$3$ mol of a mixture of $FeSO_4$ and $Fe_2(SO_4)_3$ required $100 mL$ of $2 M$ $KMnO_4$ solution in acidic medium. Hence, mole fraction of $FeSO_4$ in the mixture is:
Question 162 :
<blockquote>Amount in g of simple containing 80% NaOH required to prepare 60 litre of 0.5 M solution is :</blockquote>
Question 163 :
$NO_2$ associates as $2NO_2\rightleftharpoons N_2O_4$.The experimental molecular weight of the reaction mixture is calculated from vapour density under certain conditions was 60. The mole fraction of dimer $N_2O_4$ is:
Question 164 :
Washing soda $(Na_2CO_3.10H_2O)$ is widely used in softening of hard water. If $1$ L of hard water requires $0.0286$ g of washing soda, the hardness of $CaCO_3$ in ppm is :
Question 165 :
In $200\ g$ of a sample of olem labelled as $109.0\% ,\ 12\ g$ water is added. The new labelling of the oleum sample is
Question 166 :
A sample of oleum is labelled as $112\%$. In $200\ g$ of this sample, $18\ g$ water is added, The resulting solution will contain
Question 167 :
Calculate the mole fraction of glucose in an aqueous solution that contains 45 g of glucose in 45 g of water. Find out the weight of NaOH required to be disolved in 90 g of water in order to get a solution of the same mole fraction.
Question 168 :
${10}^{24}$ molecules of solute are dissolved in ${10}^{25}$ molecules of solvent, the mole fraction of solute in solution is :
Question 169 :
$500$ g of urea solution of mole fraction $0.2$ is diluted to $1500$ g. The mole fraction of solute in the diluted solution is:
Question 170 :
Assuming that concentration of ${Ca}^{2+}$ ions in solution is in equal equivalence ratio to chloride ions, the hardness of water is :
Question 171 :
Mole fraction of the solute in a 1.00 molal aqueous solution is :
Question 172 :
$RH_2$ (ion exchange resin) can replace $Ca^{2+}$ in hard water:       $RH_2 + Ca^{2+} \rightarrow RCa + 2H^+$<br/>$1\; L$ of hard water after passing through $RH_2$ has $pH= 2$. Hence, hardness in ppm of $Ca^{2+}$ is :<br/>
Question 173 :
A sample of water has a hardness expressed as $77.5$ ppm $Ca^{2+}$. This sample is passed through an ion exchange column and $Ca^{2+}$ is replaced by $H^+$. Select correct statement(s).<br/>
Question 174 :
Hardness in water is expressed in terms of ppm of $CaCO_3$. If hardness is $200$ ppm $CaCO_3$, then in terms of molarity, it is:<br/>
Question 175 :
A candle is burnt in a beaker until it extinguishes itself. A sample of gaseous mixture in the beaker contains $6.08\times 10^{20}$ molecules of $N_2, 0.76\times 10^{20}$ molecules of $O_2$, and $0.50\times 10^{20}$ molecules of $CO_2$. The total pressure is 734 mm of Hg. The partial pressure of $O_2$ would be:
Question 176 :
$5.5\ mg$ of nitrogen gas dissolves in $180\ g$ of water at $273\ K$ and one atm pressure due to nitrogen gas. The mole fraction of nitrogen in $180\ g$ of water at $5\ atm$ nitrogen pressure is approximately :
Question 178 :
$ V_1 $ mL of NaOH of normality x and $ V_2  mL of Ba(OH)_2 $ of normality y are together sufficient to neutralize exactly, 100 mL of $ 0.1 N HCI. If V_ 1: V_2 =  1 : 4$ and if $x : y = 4 : 1, $what fraction of the acid is neutralised by$ Ba(OH)_2 $ ?
Question 179 :
When $0.575 \times 10^{-2} kg$ of Glauber's salt ($Na_2SO_4\cdot 10 H_2O$) is dissolved in $3$ g water, we get $1$ $dm^3$ of a solution of density $1077.2 \:kg\: m^{-3}$. Calculate the molarity, molality and mole fraction of $Na_2SO_4$ in the solution:<br/>
Question 180 :
Solution of $100ml$ water contains $0.73g$ of $Mg(HCO_3)_2$ and $0.81g$ of $Ca(HCO_3)_2$. Calculate the hardness in terms of ppm of $CaCO_3$.
Question 181 :
The mole fraction of methanol in $4.5$ molal aqueous solution is:
Question 182 :
From the following data of $\Delta H$, of the following reactions,<br/>$C(s)+\dfrac{1}{2}O_2(g)\rightarrow CO(g)AH=-110$ kJ<br/>$C(s)+H_2O(g)\rightarrow CO(g)+H_2(g)\Delta H=132$ kJ<br/>What is the mole composition of the mixture of steam and oxygen on being passed over a coke at $1273$ K, keeping the temperature constant?<br/>
Question 183 :
The concentration of $\displaystyle Ca\left ( HCO_{3} \right )_{2}$ in a sample of hard water is $486$ ppm. The density of water sample is $1.0$ g/ml. The molarity of the solution is :
Question 184 :
The ppm strength of $CO_2(g)$ volume by volume (mL of $CO_2$ per $10^6$ mL of air) is:
Question 185 :
One litre of a sample of hard water contains $55.5$ mg of $CaCl_2$ and $4.75$ mg of $MgCl_2$. The total hardness in terms of ppm of $CaCO_3$ is :
Question 186 :
$30\: mL$ of $CH_{3}OH (d = 0.8\: g/cm^{3})$ is mixed with $60\: mL$ of $C_{2}H_{5}OH (d= 0.92\: g/cm^{3})$ at $25 ^\circ C$ to form a solution of density $0.88\: g/cm^{ 3}$. Select the correct option.
Question 187 :
The mole fraction of $NaCl$, in a solution containing 1 mole of $NaCl$ in 1000 g of water is:
Question 188 :
When $200\ g$ of an oleum sample labelled as $109\%$ is mixed with $300\ g$ of another oleum sample labelled as $118\%$ the new labelling of resulting oleum sample becomes
Question 189 :
$1$ mole $N_{2}$ and $4$ mole $H_{2}$ are allowed to react in a vessel and after reaction $H_{2}O$ is added to the vessel. Aqueous solution required $1$ mole $HCl$. Mole fraction of $H_{2}$ in the gaseous mixture after the reaction is:
Question 190 :
What is the percentage of carbon, by wt. in vitamin $C$ ?
Question 191 :
Units of parts per million (ppm) or parts per billion (ppb) are often used to describe the concentrations of solutes in very dilute solutions. The units are defined as the number of grams of solute per million or per billion grams of solvent. Bay of Bengal has $1.89$ ppm of lithium ions. The molality of $\displaystyle Li^{+}$ in this water is (atomic number of $Li=7$) :
Question 193 :
$12.8$ g of mixture of $MCl$ (volatile) and $KCl$ (non-volatile) on reaction with excess of aqueous $AgNO_3$ solution gave $28.7$ g white precipitate. $12.8$ g of same mixture on a heating gave a gas that on passing into $AgNO_3$ solution gave $14.35$ g of white precipitate. Hence :<br/>
Question 194 :
The hardness of water due to $HCO^{\circleddash}_3$ is $122$ ppm. Select the correct statement(s).<br/>
Question 195 :
The compound $Na_2 CO_3 . x H_2O$ has $50  \%  H_2O$ by mass. The value of "x" is :
Question 196 :
What is the percentage of hydrogen by wt.in vitamin $C$ ?
Question 200 :
Maximum number of covalent bonds formed by N and P respectively are :
Question 201 :
Which of the following pairs is obtained on heating ammonium dichromate?
Question 204 :
Nitrogen form $N_2$, but phosphorous form $P_2$, it'sat a time convert in $P_4$, reason is : -
Question 207 :
The type of bond between the atoms in a nitrogen molecule is :
Question 208 :
Which of the following is correct statement regarding $N(CH_3)_3$ and $N(SiH_3)_3$?
Question 209 :
Which of the following oxides of nitrogen are coloured in nature?
Question 213 :
Which one of the following elements does not form the compound, M$_{4}$ O$_{10}$ (M = element)?<br/><br/>
Question 216 :
Which of the following is correct about V group Hydrides (from ammonia to Bismuthine)<br/>
Question 219 :
Which of the following is an example of super octet molecule?
Question 222 :
Assertion: $H_{3}PO_{4}$ and $H_{3}PO_{3}$ both are present in fertilizers.
Reason: $H_{3}PO_{3}$ increases the solubility of fertilizers.
Question 226 :
The impurities formed during laboratory preparation of dinitrogen from ammonium chloride and sodium nitride is/are:
Question 227 :
Nitrogen differs from P, As, Sb and Bi in the following properties :
Question 231 :
Give examples of compounds of N with oxidation number -3 to +5.
Question 232 :
Assertion: Catenation tendency is weaker in nitrogen.
Reason: Nitrogen exists as diatomic gas.
Question 233 :
Match List I with List II and select the correct answer using the codes given below the lists<table class="wysiwyg-table"><tbody><tr><td>List I (Compound)</td><td>List II (Oxidation state of $N$)</td></tr><tr><td>(A) ${NO}_{2}$</td><td>(1) $+5$</td></tr><tr><td>(B) $HNO$</td><td>(2) $-3$</td></tr><tr><td>(C) ${NH}_{3}$</td><td>(3) $+4$</td></tr><tr><td>(D) ${N}_{2}{O}_{5}$</td><td>(4) $+1$</td></tr></tbody></table>
Question 237 :
In which of the following arrangement the order is not according to the property indicated against it:
Question 238 :
Which of the following compounds do not exist? <br/><br/>$N_4, P_4, PCI_5, NCI_5, P_2O_5, NO_2, PO_2$ :
Question 239 :
In Nitrogen family the H-M-H angle in the hydrides $MH_{3}$ gradually becomes closer to $90^0$ on going from N to Sb. This shows that gradually :<br/>
Question 240 :
Of the following hydrides which one has the lowest boiling point?
Question 245 :
Trimethyl phosphine oxide can be represented by two canonical forms as shown but trimethylamine oxide can't be. It is due to:<br/>
Question 247 :
Assertion: Nitrogen gas is filled in electric bulbs.
Reason: Nitrogen is colorless, odorless and tasteless gas.
Question 248 :
Pentavalence in phosphorus is more stable when compared to that of nitrogen even though they belong to same group is due to :
Question 249 :
$N{ F }_{ 3 }$ is stable. But, $N{ Cl }_{ 3 }$ is explosive. Identify the reason :
Question 250 :
Which of the following halide of nitrogen is not explosive in nature?
Question 252 :
Which of the following compound is used in detection of metal ions such as $Cu^{+2},\ Ag^{+}$?
Question 253 :
Nitrogen has unique ability to form $\displaystyle P\pi -P\pi $ multiple bonds with itself and with other elements due to:
Question 254 :
Which forms $p\pi - p \pi$ multiple bonds with itself and with C and O?
Question 255 :
$N{ F }_{ 3 }$ is stable, but $N{ Cl }_{ 3 }$ is explosive. Identify the reason.
Question 258 :
Assertion: A mixture of nitrogen and carbon dioxide can be distinguished by passing it through lime water.
Reason: Nitrogen does not turn lime water milky.
Question 259 :
In atoms of nitrogen molecule, which type of bond are present ?<br/>
Question 263 :
The Group VA element that makes up part of bones and teeth is:<br/>
Question 264 :
In which of the following compounds the oxidation state of nitrogen is most negative?
Question 265 :
$CN^-$ and $N_2$ are isoelectronic. But in contrast to $CN^-, N_2$ is chemically inert because of:
Question 268 :
The element which forms oxides in all oxidation states + 1 to + 5 is:
Question 269 :
Which is in the decreasing order of boilingpoints of $\mathrm{V}$ group hydrides?<br>
Question 270 :
Which of the following oxoacids of nitrogen has most acidic character?<br/>$I.\ N_2O_3 \\ II.\ NO_2 \\ III. \ N_2O_5 \\ IV.\ NO$
Question 274 :
Assertion: Assertion: $N_2$ is less reactive than $P_4$
Reason: Reason: Nitrogen has more electron gain enthalpy than phosphorus.
Question 276 :
Nitrogen forms stable $N_{2}$ molecule but phosphorus is converted to $P_{4}$ from $P_{2}$because:
Question 278 :
Which of these ions need to be stored in fairly acidic media?
Question 279 :
The oxides of $15^{th}$ group elements of the type $M_{2}O_{3}$ which will exhibit amphoteric or basic properties but no acidic properties are__________.<br/>
Question 280 :
The boiling point of the following hydrides follow the order of
Question 281 :
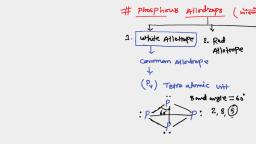
Assertion: Elementary phosphorus exists in three principal allotropic forms, i.e. white (or yellow), red (or violet) and black
Reason: Of the three forms, white phosphorus is the most important and most reactive<br/>Read the above assertion and reason and choose the correct statement regarding it.<br/>
Question 282 :
Which of the following halide of nitrogen is stable in nature?
Question 285 :
The Lewis base strength of nitrogen halide is the maximum for :<br/>
Question 287 :
Nitrogen and phosphorus are both in Group 15 of the Periodic Table. Phosphorus forms a chloride with the formula $P{Cl}_{5}$ but nitrogen does not form $N{Cl}_{5}$.<br>Which statements help to explain this?<br>1. Nitrogen's outer shell cannot contain more than eight electrons.<br>2. Nitrogen cannot have an oxidation state of $+5$<br>3. Nitrogen is less electronegative than phosphorus.
Question 289 :
The reactivity of the following compounds toward water is in the order:
Question 291 :
Assertion: Nitrogen is unreactive at room temperature but becomes reactive at elevated temperatures or in presence of a catalyst.
Reason: In nitrogen molecule, there is extensive delocalization of electrons.
Question 294 :
In case of nitrogen, $ NCl_3$ is possible but not $NCl_5$ whereas in case of phosphorus $PCl_3$ as well as $PCl_5$ are possible. It is due to:
Question 298 :
When bismuth chloride is poured into a large volume of water, the white precipitate formed is: