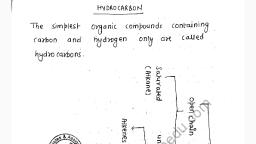
Question 1 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">In a galvanic cell, the reactions taking place in the anodic half cell and the cathodic half cell will be:</p>
Question 4 :
KCl can be used in salt bridge as electrolyte in which of the following cells?
Question 6 :
In a cell, the following reaction takes place: $Fe + CuSO_{4} \rightarrow FeSO_{4} + Cu$. The cell can be represented as:
Question 7 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Hydrogen -Oxygen fuel cells are used in space craft to supply:</p>
Question 8 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Which of the following energy changes occur in galvanic cell?</p>
Question 9 :
When the electric current is passed through a cell having an electrolyte, the positive ions move towards cathode and negative ions towards the anode. If the cathode is pulled out of the solution then____________.
Question 10 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">The potential across the metal and the aqueous solution of its ions of unit activity at 298K is known as:</p>
Question 11 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">In which reactants are not contained within the cell but are continuously supplied from external source?</p>
Question 12 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Which of the following reaction is used to make a fuel cell ?</p>
Question 13 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Which one of the following cells can convert chemical energy of $O_{2}$ and $H_{2}$ directly into electrical energy:</p>
Question 18 :
Assertion: Dry cell is a primary cell and lead storage cell is a secondary cell.<br/>Reason: A primary cell can only be used once while secondary cells are rechargeable.<br/>
Question 20 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Following are some of the facts about dry cell</p><p class="wysiwyg-text-align-left">i) It is also called Leclanche cell</p><p class="wysiwyg-text-align-left">ii) It is also called Daniel cell</p><p class="wysiwyg-text-align-left"><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">iii) Electrolyte is a moist paste of NH$_{4}$Cl<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">and ZnCl$_{2}$<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">in starch</p><p class="wysiwyg-text-align-left">iv) Cathodic reaction is</p><p class="wysiwyg-text-align-left"><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">MnO<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">$_{2}$<span class="wysiwyg-font-size-xx-small"><span class="wysiwyg-font-size-xx-small"> <span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">(s) + NH$_{4}^{+}$<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"> + e$^{-}\rightarrow $<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">MnO(OH)+NH$_{3}$</p><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p>Select correct facts :</p>
Question 21 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Lithium is generally used as an electrode in high energy density batteries. This is because:</p>
Question 22 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Which of the following statements is true for fuel cells ?</p>
Question 25 :
State True or False.<br/>The cell voltage is a quantitative measure of the driving force of the cell reaction.
Question 26 :
Assertion: In a Daniell cell, electrons flow from zinc electrode to copper electrode outside the cell.Reason: In Daniell cell, Zn electrode is anode and Cu is cathode.<br/>
Question 27 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Daniel cell can be represented in which of the following way?</p>
Question 28 :
Assertion: To obtain maximum work from a galvanic cell charge has to be passed reversibly.
Reason: The reversible work done by a galvanic cell is equal to decrease in its Gibbs energy.
Question 29 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">Some of the batteries have alkaline electrolytes-</p> <p class="wysiwyg-text-align-left">i) Nickel - cadmium ii) Mercury battery</p> <p class="wysiwyg-text-align-left">iii) Modified Leclanche cell</p> <p><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">Cell potential is independent of [OH$^{-}$<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">] in :</p>
Question 30 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p>The life span of a Daniel cell may increased by:</p>
Question 31 :
<p class="wysiwyg-text-align-left"><span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">The metal which cannot liberate $H_2$<span class="wysiwyg-font-size-xx-small"><span class="wysiwyg-font-size-xx-small"> <span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small">gas from hydrochloric acid is:</p>
Question 33 :
The weights of various ions liberated by passing the same current through different electrolytes are in the ratio of their:
Question 34 :
<font color="#333333">State True or False.</font>In an electrode concentration cell, the cell reaction Zn(C$_1$) $\rightarrow$ Zn(C$_2$) will be spontaneous if c$_1$  > c$_2$ .
Question 36 :
Assertion: The anions migrate to the cathode in an electrochemical reaction
Reason: Positively charged ions are attracted to the negatively charged electrode
Question 37 :
If the size of the electrodes is increased, the corresponding cell voltage will:<br/>
Question 38 :
Assertion: The electrolysis of potassium iodide, KI, produces electrical energy.
Reason: Electrolytic cells convert chemical energy into electrical energy
Question 39 :
The value of equilibrium constant for a feasible cell reaction is:
Question 40 :
The oxidation potential of a hydrogen electrode at $pH = 10$ and $P_{H_2}$ = 1 atm is:
Question 42 :
An electrolytic cell is constructed for preparing hydrogen. For an average current of 1 ampere in the circuit, the time required to produced $450mL$ of hydrogen at NTP is approximately.
Question 43 :
Which of the following statements are correct about voltaic cell?<br>(I) Chemical energy isconverted into electrical energy.<br>(II) Electrons flow from anode to cathode.<br>(III) Oxidation occurs at anode.<br>(IV) Anode is negative and cathode is positive.
Question 44 :
A reversible galvanic cell is connected to an external battery. If the EMF of the battery is less than the EMF of the galvanic cell, then the current:
Question 45 :
Two platinum electrodes were immersed in a solution of $CuSO_4$ and electric current was passed through the solution. After some time, it was found that colour of $CuSO_4$ disappeared with evolution of gas at the electrode. The colorless solution contains:<br/>
Question 46 :
What will be the emf of a cell of two hydrogen electrodes, if a negative electrode is in contact with $10^{-8}M\ H^+$ion positive electrode is in contact with $0.025M\ H^+$?
Question 47 :
Assertion: When an electric current is passed through an aqueous solution of sodium chloride, sodium ions move towards cathode and chloride ions move towards anode.
Reason: Cathode is a negatively charged electrode and therefore attracts Na$^+$ ions. Anode is a positively charged electrode and therefore attracts Cl$^-$ ions.
Question 48 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">In a Daniel cell when $Cu$ and $Zn$ electrodes are connected current flows from:</p>
Question 50 :
Write true or false for the following statements :<br>A solution of an ionic compound in water contains ions , which move to the opposite electrodes when electricity is passed through the solution.
Question 51 :
The purpose of a salt bridge in a galvanic cell is to:
Question 55 :
Which of the following will increase the voltage of the cell? <br/><br/>$Sn(s) + 2 \text{Ag}^+ (aq) \rightarrow 2Ag(s) + Sn^{2+}$
Question 56 :
During electrolysis of fused calcium hydride, the hydrogen is produced at:
Question 57 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">If a salt bridge is not used between two half cells, what happens to the voltage?</p>
Question 58 :
In a cell that utilizes the reaction $Zn(s)$ $+2\mathrm{H}^{+}$ (aq) $\rightarrow \mathrm{Z}\mathrm{n}^{2+}(\mathrm{a}\mathrm{q})+\mathrm{H}_{2}(\mathrm{g})$ addition of $\mathrm{H}_{2}\mathrm{S}\mathrm{O}_{4}$ to cathode compartment, will:  <br/>
Question 59 :
The chemical reaction, $2AgCl_{(s)} + H_{2(g)} \rightarrow 2HCl_{(aq)} + 2Ag_{(s)}$ taking place in a galvanic cell is represented by the notation:
Question 60 :
<span class="wysiwyg-font-size-small"><span class="wysiwyg-font-size-small"><p class="wysiwyg-text-align-left">In a galvanic cell, the positive ions of salt bridge migrate towards:</p>
Question 61 :
<p>Given standard $E^{\ominus}$:</p><p>$Fe^{3+}\, +\, 3e^-\, \rightarrow\, Fe$; $E^{\ominus}\, =\, -\, 0.036\, V$ </p><p>$Fe^{2+}\, +\, 2e^-\, \rightarrow\, Fe$; $E^{\ominus}\, =\, -\, 0.440\, V$ </p><p>The $E^{\ominus}$ of $Fe^{3+}\, +\, e^-\, \rightarrow\, Fe^{2+}$ is:</p>
Question 63 :
Among $Na$, $Hg$, $S$,$ Pt$, and graphite which can be used as electrodes in electrolytic cell having aqueous solutions ?
Question 64 :
Which of the following reactions is used to make a fuel cell?
Question 66 :
<font color="#333333">State True or False.</font>In a galvanic cell, the half-cell with higher reduction potential acts as a reducing agent.