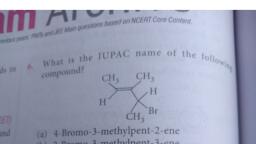
Question 1 :
Linear combination of two hybridised orbitals belonging to two atoms and each having one electron leads to the formation of:
Question 3 :
The hybrid orbitals with 25% s character is involved in formation of ?
Question 4 :
Compounds which contain oxides of carbon or carbonate hydrogencarbonate ions are called:
Question 5 :
Which of the following reagents maybe used to distinguish between but-2-yne and but-1-yne.<br/>
Question 11 :
Assertion: Why organic compounds are studied separately as  'Organic Chemistry'.
Reason: There is a huge number of compounds of carbon due to its catenation and multiple bond formation property.
Question 12 :
The percentage of p-character in the orbitals forming $P-P$ bonds in $P_4$ is:
Question 13 :
The conversion of $ - \operatorname { coch } _ { 2 } - t o - C H _ { 2 } - C H _ { 2 } - $ using Hg/ Zn and HCI is known as
Question 14 :
If true enter 1, else enter 0.<br>The $H-N-H$ bond angle in ${NH}_{3}$ is greater than $H-As-H$ bond angle in $As{H}_{3}$.
Question 18 :
What is the percentage of p-character of hybrid orbitals of carbon in methane, ethene and ethyne respectively?
Question 19 :
<p>Which of the following statements are usually correct for carbon compounds? </p><p>These :</p><p>(a) are good conductors of electricity.</p><p>(b) are poor conductors of electricity.</p><p>(c) have strong forces of attraction between their molecules.</p><p>(d) do not have strong forces of attraction between their molecules.</p>
Question 21 :
What is the decreasing order of strength of the given\  bases?<br/>${ OH }^{ - },{ NH }_{ 2 }^{ - },\ H-C\equiv { C }^{ - }$ and ${ CH }_{ 3 }-{ CH }_{ 2 }^{ - }$
Question 24 :
The $Cl-C-Cl$ angle in $1,1,2,2$ - tetrachloroethene and tetrachloromethane respectively will be about:
Question 26 :
Assertion: The bond angle of $P{Br}_{3}$ is greater than ${PH}_{3}$ but the bond angle of $N{Br}_{3}$ is lesser than ${NH}_{3}$.
Reason: Electronegativity of $P$-atom is less than that of $N$-atom.
Question 28 :
The decreasing value of bond angles from $NH_3$ ($106^{\circ}$) to $SbH_3$ ($101^{\circ}$) down group-15 of the periodic table is due to:<br/>
Question 30 :
Bond angle between two hybrid orbitals is ${105}^{o}$. Hybrid character orbital is: