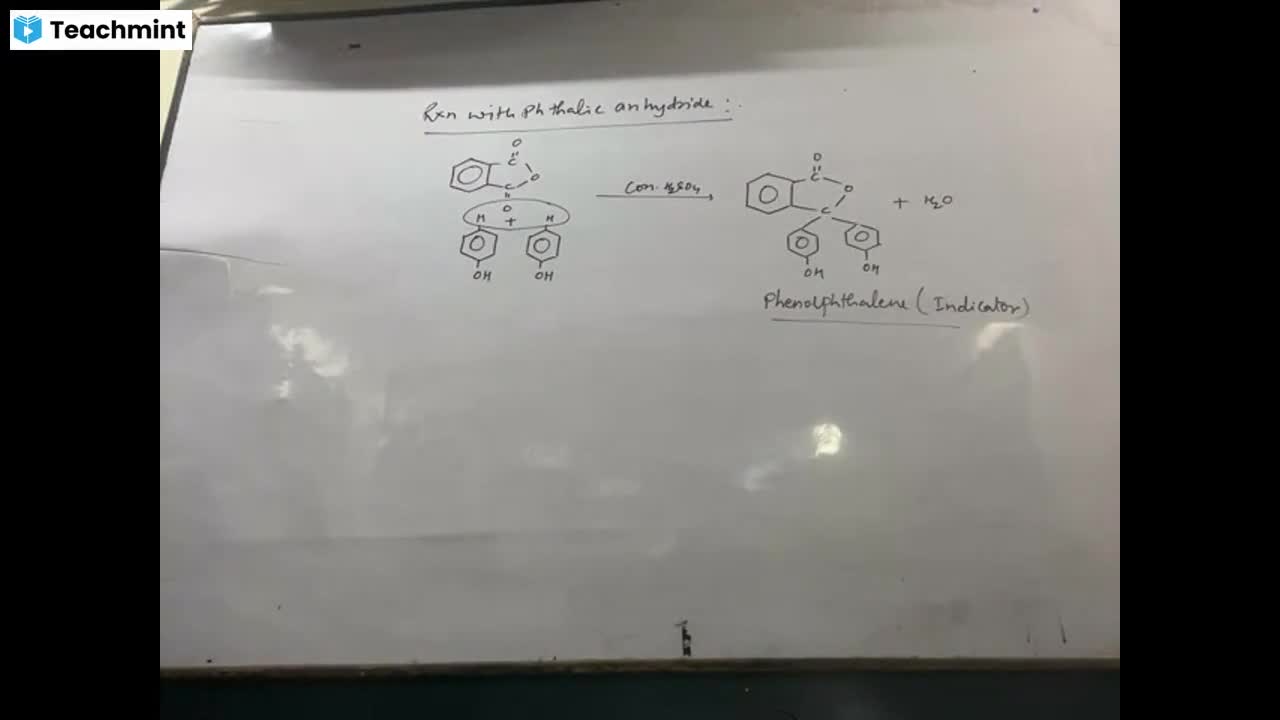
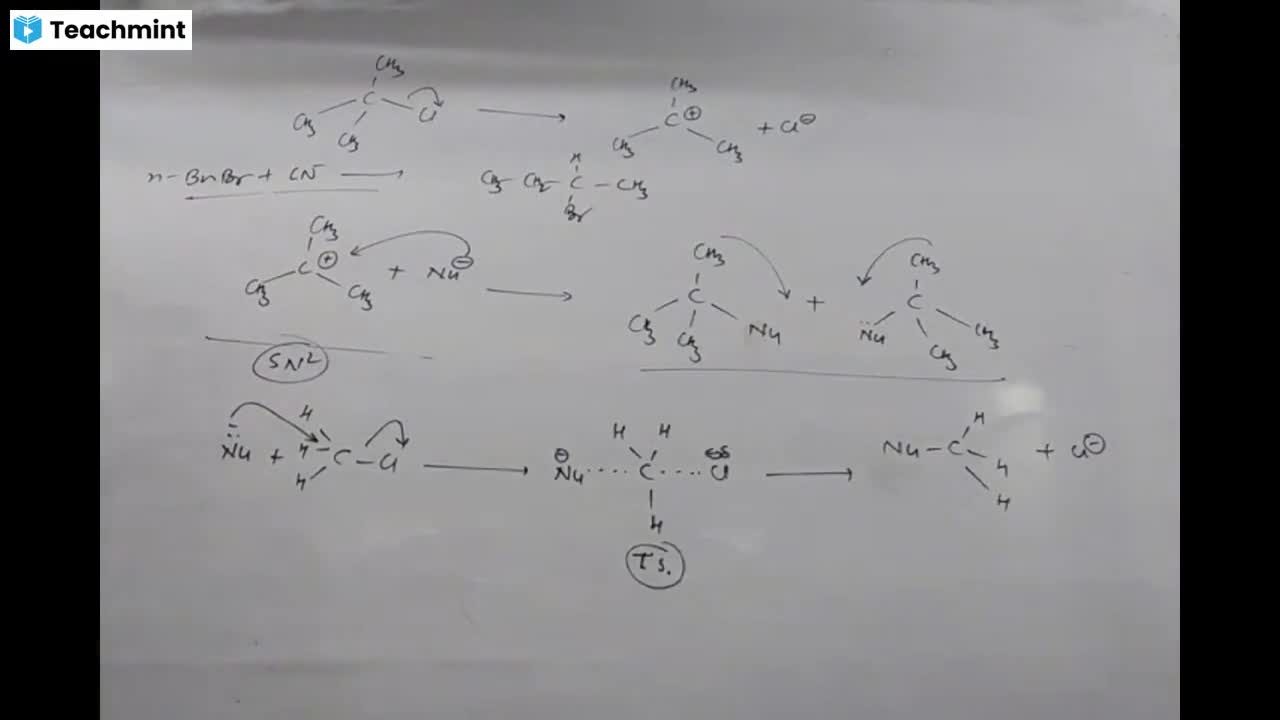
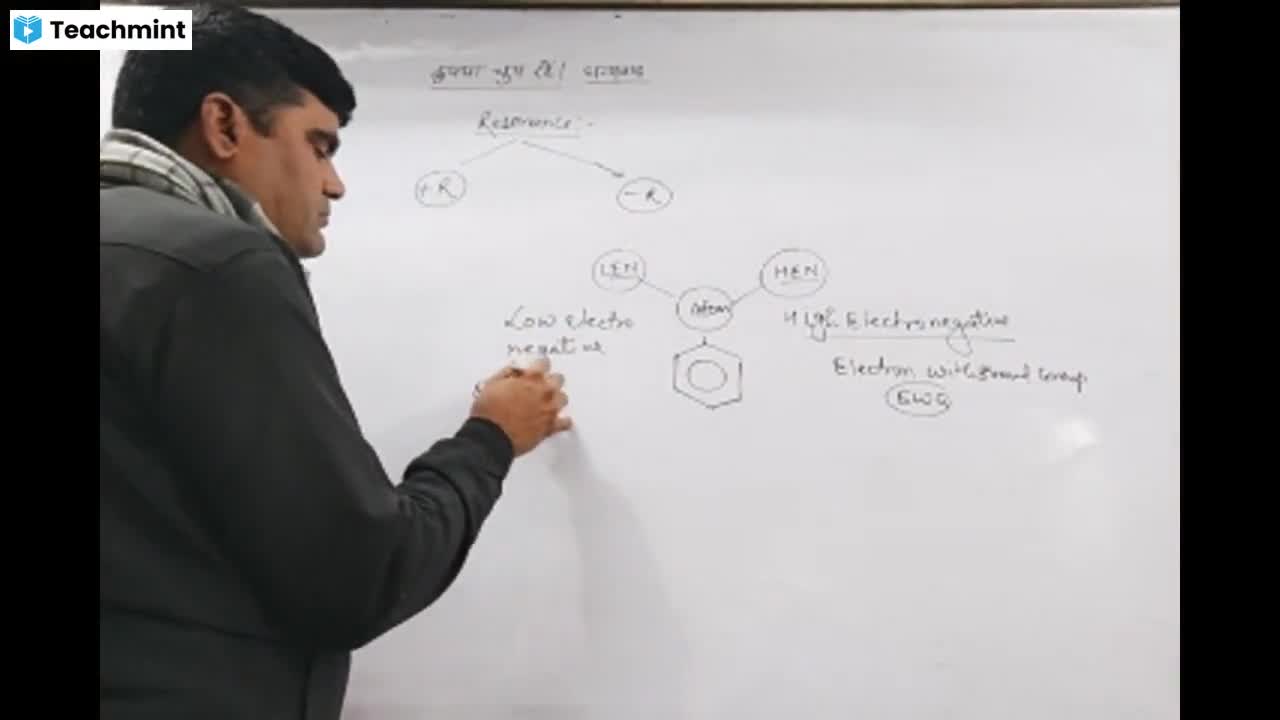
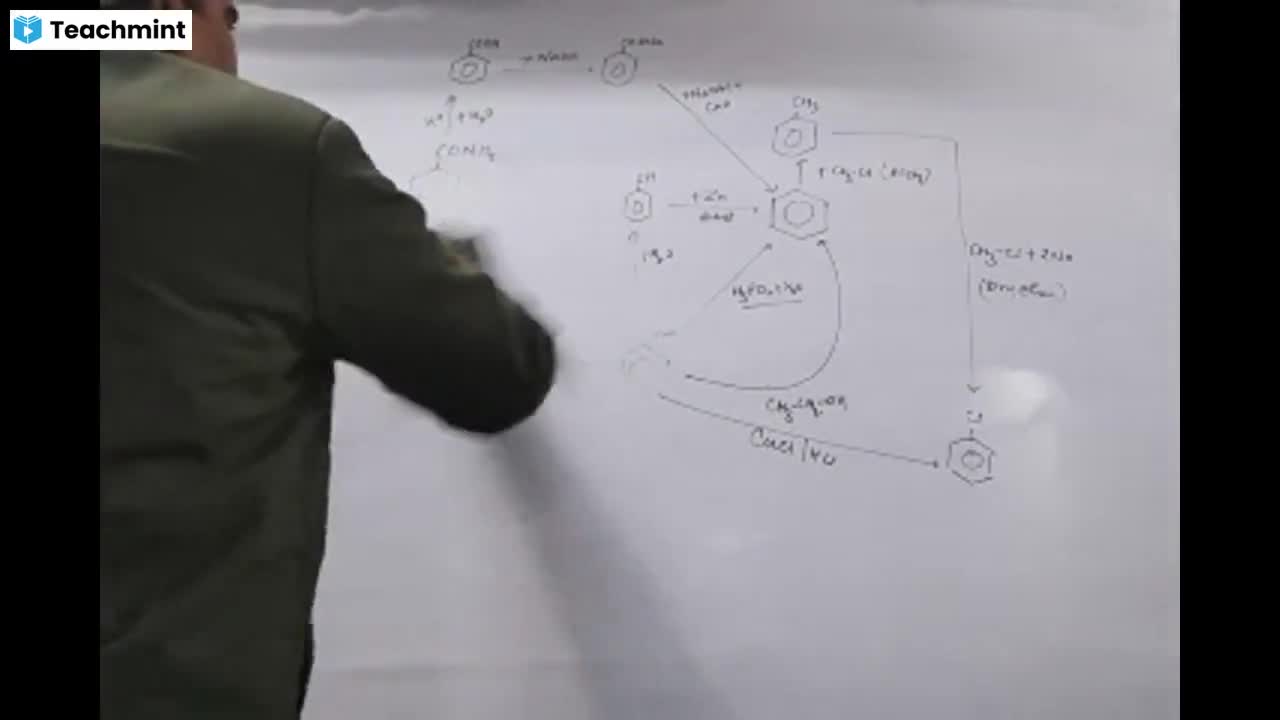
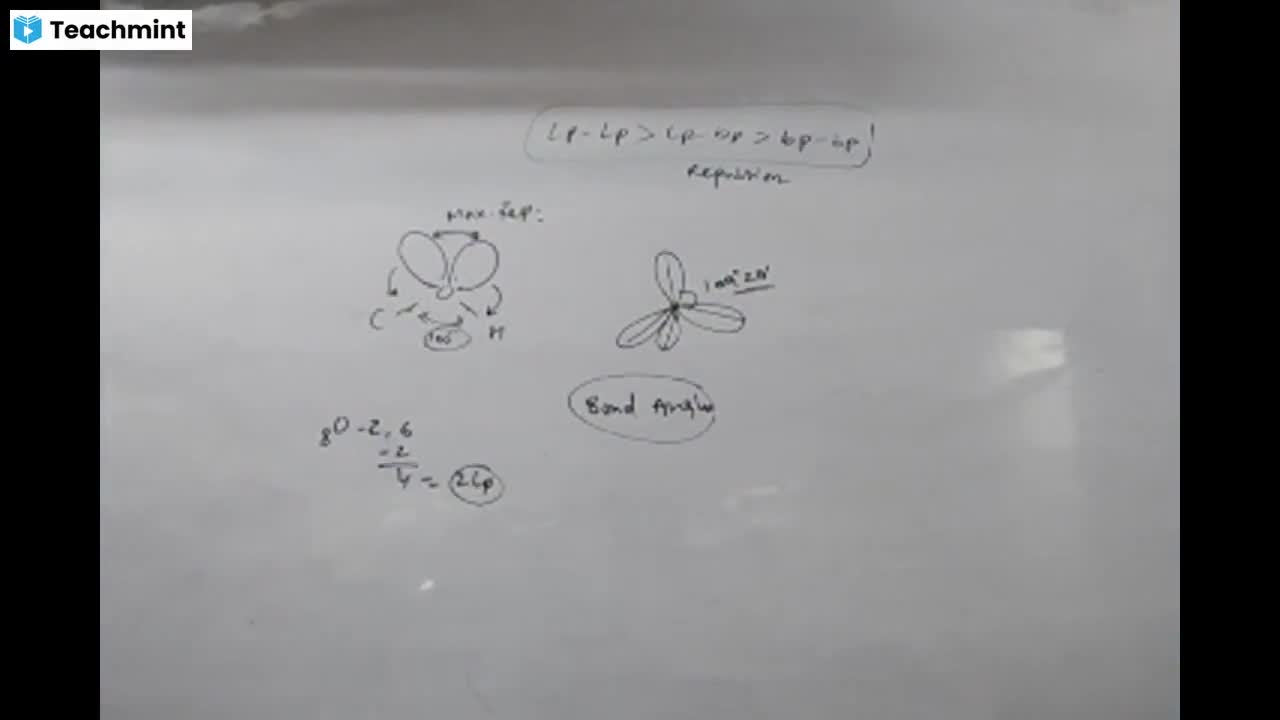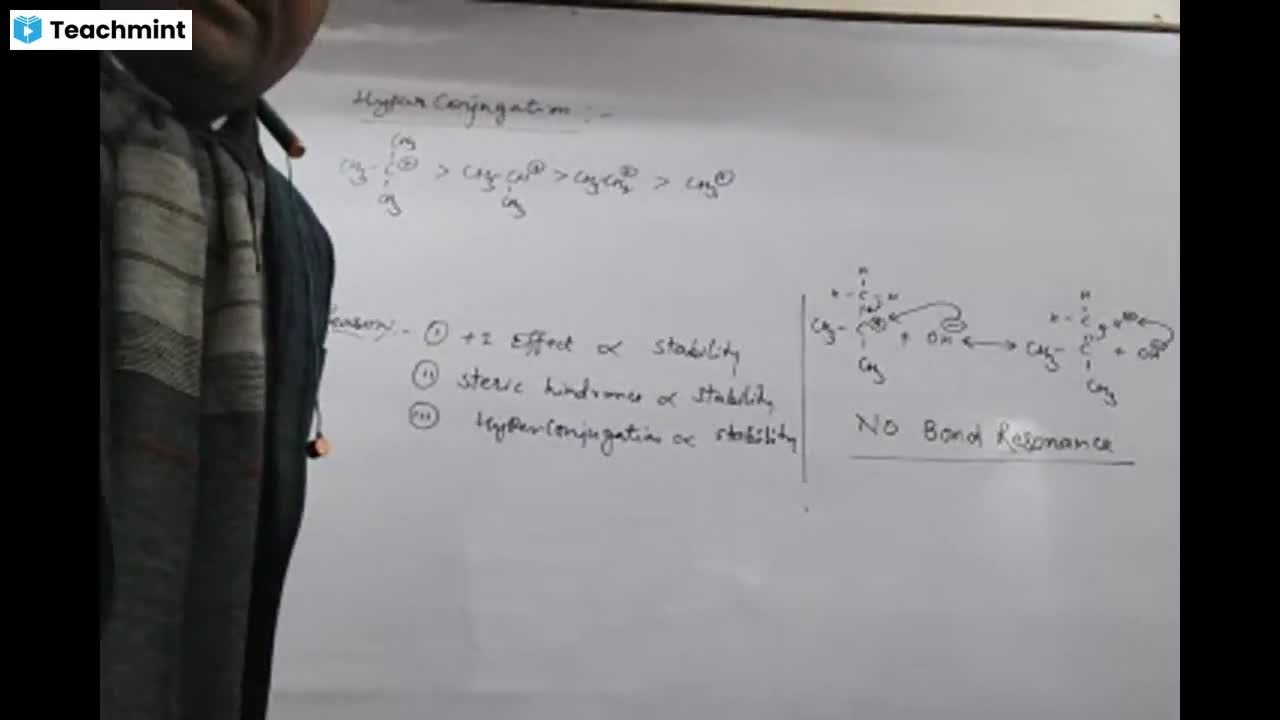Question 1 :
During the oxidation of $Mn^{2+}$ to $MnO_{4}^{-}$ by $PbO_{2}$ in acid medium, the number of moles of acid consumed per mole of $Mn^{2+}$ ion is
Question 3 :
What is the oxidising agent in the following reaction?<br>$2Fe^{2+}_{aq} + Cl_{2(g)} \rightarrow 2Fe^{3+}_{(aq)} + 2Cl^-_{(aq)}$
Question 5 :
When the equation: $C_2H_6 + O_2 \rightarrow CO_2 + H_2O$ is completely balanced using the lowest whole number coefficients, the sum of the coefficients will be :
Question 6 :
Statement 1: The $Cu^{2+}$ ion needs to be oxidized to form Cu metal.<br/>Statement 2: Oxidation is a gain of electrons.
Question 7 :
Which of the following statements about the given reaction are correct ?<br>$3Fe(s) + 4H_{2}O(g) \rightarrow Fe_{3}O_{4}(s) + 4H_{2}(g)$<br>(i) Iron metal is getting oxidised.<br>(ii) Water is getting reduced.<br>(iii) Water is acting as reducing agent.<br>(iv) Water is acting as oxidising agent.<br>
Question 8 :
Which of the following statement is correct for the oxidation reaction?
Question 9 :
In the process of burning of magnesium in air, magnesium undergoes :
Question 10 :
When Magnesium is burnt in air, a white ash remain as left over. What is this?
Question 11 :
For joining railway tracks, ferric oxide is reacted with aluminium to produce aluminium oxide and iron. The balanced chemical equation for the given reaction is<br>$\underset {Ferric\ oxide}{Fe_2O_{3(x)}}+\underset {Aluminium}{2Al_{(x)}}\rightarrow \underset {Aluminium\ oxide}{Al_2O_{3(x)}}+\underset {Iron}{2Fe_{(x)}}$<br>Which of the following substances is oxidized in the given reaction?<br>
Question 12 :
The substance oxidized and the oxidizing agent in the following reaction are respectively :<br/>$Zn+H_2SO_4\rightarrow ZnSO_4+H_2$
Question 15 :
In the given reaction,<br>$2{ K }_{ 4 }\left[ Fe{ \left( CN \right) }_{ 6 } \right] \left( aq \right) +{ H }_{ 2 }{ O }_{ 2 }\left( aq \right) \longrightarrow \\ 2{ K }_{ 3 }\left[ Fe{ \left( CN \right) }_{ 6 } \right] \left( aq \right) +{ 2KOH }\left( aq \right) $<br>Which of the following processes takes place?
Question 17 :
Consider the following reaction, taking place in a container fitted with a movable piston.<br>$2SO_{2}(g) + O_{2}(g) \rightarrow 2SO_{3}(g)$<br>Suppose we place two moles each of $SO_{2}$ and $O_{2}$ in the reaction vessel at $25^{\circ}C$, and adjust the volume to give a total pressure of $1.0\ atm$. The reaction is ignited by a spark, and goes to completion. The temperature is returned to $25^{\circ}C$.<br>Which of the following best describes this system after reaction is complete?
Question 19 :
State whether the given statement is true or false.<br/>$X^{2+} + 2e^{-} \rightarrow X$ is an example of oxidation.
Question 24 :
In the reaction,<br/><br/>$3CuO+2{ NH }_{ 3 }\longrightarrow { N }_{ 2 }+3{ H }_{ 2 }O+3Cu$<br/><br/>the change of ${NH}_{3}$ to ${N}_{2}$ involves:
Question 25 :
For the galvanic cell, $Cu|Cu^{2+}||Ag^+|Ag$. Which of the following observations is not correct?
Question 27 :
_______ is a degradation phenomenon in which the electrons in a plastic are so strongly attracted to oxygen atoms.
Question 28 :
On the basis of the following ${E}^{o}$ values, the strongest oxidising agent is:<br>${ \left[ Fe{ \left( CN \right) }_{ 6 } \right] }^{ 4- }\longrightarrow { \left[ Fe{ \left( CN \right) }_{ 6 } \right] }^{ 3+ }+{ e }^{ - },{ E }^{ o }=-0.35V$<br>${Fe}^{2+}\rightarrow {Fe}^{3+}+{e}^{-1}$;<br>${Fe}^{2+}\rightarrow {Fe}^{3+}+{e}^{-1};{E}^{o}=-0.77V$
Question 29 :
Consider the following $E^{0}$ values:<br/>$E_{Fe^{3+}/Fe^{2+}}^{0}$ = 0.77V,<br/>$E_{Sn^{2+}/Sn}^{0}$ = -0.14V<br/>Under standard conditions the potential for the reaction, is:<br/>$Sn(s)+2Fe^{3+}(aq)\rightarrow 2Fe^{2+}(aq)+Sn^{2+}(aq)$
Question 31 :
In a reaction, $4$ moles of electrons are transferred to $1$ mole of $HNO_3$ when acted as an oxidant. The possible reduction product is:
Question 32 :
When an alkali metal is reacted with hydrogen then metallic hydride is formed. In this reaction:
Question 33 :
Consider the reaction: $Cl_{2} + 2KI \rightarrow I_{2} + 2KCl$<br>Which species will be reduced at the cathode?
Question 36 :
For the reaction :<br>$ I^- + Cl{ O }_{ 3 }^{ - } + H_2SO_4 \rightarrow Cl^- + HS{ O }_{ 4 }^{ - } + I_2 $<br>
Question 37 :
Which of the following statements is not correct about the given reaction ?<br>$K_4[Fe(CN)_6 \xrightarrow{Oxidation} Fe^{3+} + CO_2 + N{ O }_{ 3 }^{ - }$
Question 40 :
Which of the following species has been oxidized?<br>$Mg(OH)_{2(aq)} + 2Na_{(s)} \rightarrow Mg_{(s)} + 2NaOH_{(aq)}$
Question 41 :
A student places a strip of solid magnesium metal into a beaker full of $100.mL$ of $3.0$ M hydrochloric acid solution. The student observes bubbling and the disappearance of the magnesium metal.<br>Which species is being oxidized and which species is being reduced in this reaction?
Question 43 :
Identify tthe compounds which are reduced and oxidised in the following reaction.<br>$3N_2H_4 + 2Br{ O }_{ 3 }^{ - }\rightarrow 3N_2 + 2Br^- + 6H_2O$
Question 44 :
In the reaction : $Cl_2 + OH^- \rightarrow Cl^- +Cl{ O }_{ 4 }^{ - }+H_2O$ ?<br/>