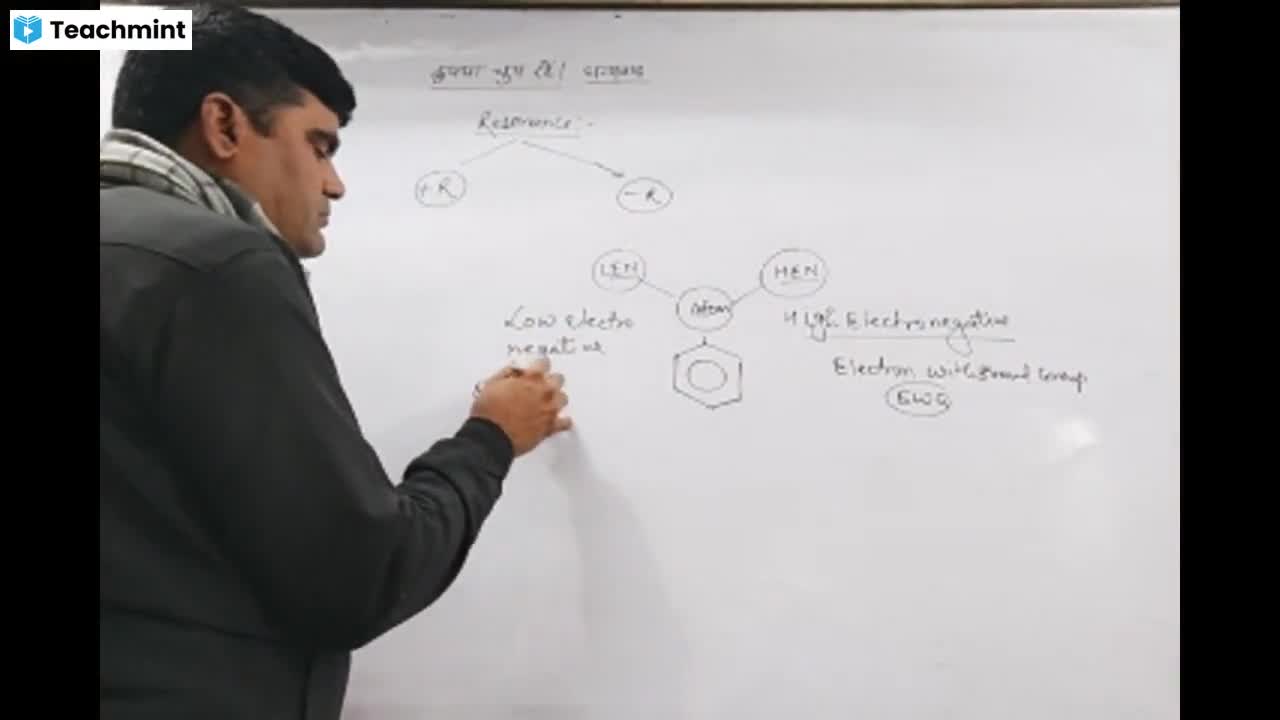
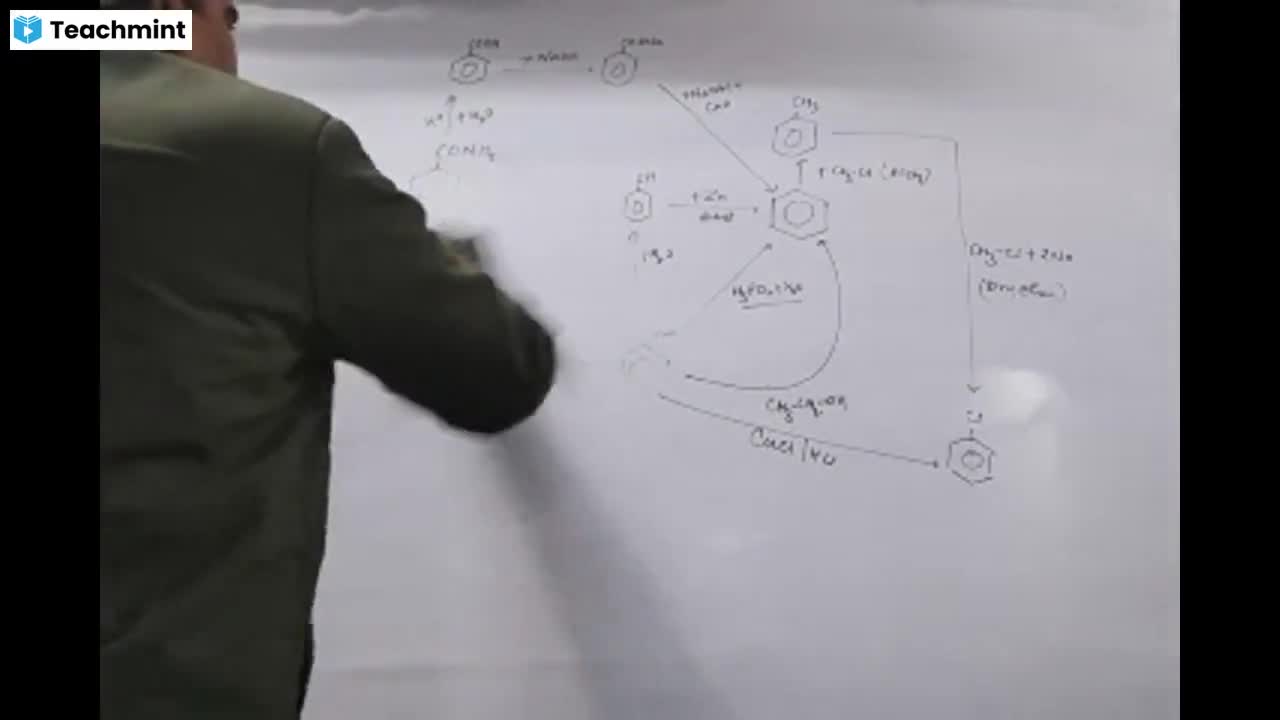
Page 1 :
New. Dimension Institute, satyaveer sir, , 12., 13., , 14,, , Oxidation is defined as;, , {a) loss of electrons (b) gain of clectrons, {c) gain of protons (a) loss of protons, The reaction, HyS + HyO; = S + 211)O manifests:, (a) oxidising action of 1,0, , (b) reducing nature of 10), , (eo) acidic nature of Hy Oy, , (d) alkaline nature of Hy Oy, , The oxidation number of Fe in Ky Fe(CN) , is?, , (a) +6 (b) +4, , {c) +3 (i) +2, , In NKCO),, the oxidation state of Ni is;, , (a) 4 (b) zero, , (c) 2 (qd) 8, , The oxidation number of carbon in CHO is:, , @) 4 (b) +2 (c) zero (d) ++, , When P reacts with caustic soda, the products are PH; and, , Nall? PO ¢ The reaction is an example of:, , {a) oxidation, , (b) reduction, , (c) bouw oxidation and reduction, , (d) neutralisation, , Which one of the following is a redox reaction?, , (9) H, + Br, = 2HBr, , (b), 2NaCl + H»SO, = NajSO, + 2HCL, , (c) HCl + AgNO, =AgCl+ HNO,, , (a) NaOH + HCI = NaCl + HO, , Which of the following is not a redox change?, , (a) 2H)S + SO; = 2H,0+3S, , (b) 2Ba0 + 0) = 2Ba0,, , (c) BaQ, + H>SO, = BaSO, + HO;, , (d) 2KCIOy = 2KCl +30), , The conversion of KzCr2O) into Cr) (SO4)q is a process, of:, , (a) oxidation (b) reduction, , (c) decomposition (d) substitution, , In the reaction, 2Na2S,03 +12 =Na 2540, + 2Nal, Tp, acts as:, , (a) reducing agent, , (b) oxidising agent, , (c) oxidising as well as reducing agent, , (d) none of the above, , Select the compound in which chlorine shows oxidation, , _ State +7?, (a) HCIO, (b) HCIO,, (c) HCIO, (d) HCIO, , Which one of the following is a reducing agent?, (a) Ozone (b) Chlorine (c) FeCl, — (d) Na2SO;, The oxidation number of nitrogen in NII; OH is:, (a) zero (hy +1 (c) -1 (ud) -2, HBr and »Hlereduce sulphuric acid. HCI can reduce, , KMnOg and, HP can reduce:, (a) Hy80 (b) KMnO,, (ce) K3CHO) (d) none of these, , 16,, , 17., , 19,, , 20,, , 21., , 22., , 24., , 25., , 26,, , Page | of 2, , | One mole of Ny Hy loses 10 mole of electrons to form, , hew compound Y. Assuming that all nitrogen appear in, new compound, what is the oxidation state of nitrogsey, (There is no change in the oxidation state of hydrogen.), {a) =I (b) -3 (c) +3 (d) +5, The brown ring complex compound is formulated as, [Fe(H0)s(NO)|SOq. The oxidation state of iron is:, (a) 1 (b) 2 () 3 (4) zero, A solution of sodium metal in liquid ammonia is strongly, reducing due to the presence of;, (a) sodium atoms (b) sodium hydroxide, (c) sodium amide (d) solvated electrons, In which of the following compounds, iron has lowest, oxidation state?, (a) FeSO, -(NH4)2 SO, -6H20, (b) KgFe(CN)¢, (c) FepogO, (d) Fe(CO)s;, When KMnQq is reduced “with oxalic acid in acidic, solution, the oxidation nwetiber of Mn. changes from:, (a) Tto2 (b) 704 (ch 76 (d) 6t0 2°, In the following reactiony :, , 3Br, +6CO}- +3H,0=5Br” + BrO} + 6HCO;, {a) bromine is oxidised, carbonate is reduced, (b) bromine is'reduced, carbonate is oxidised, {c) bromine iis néither reduced nor oxidised, {d) bromine is rediiced as well as oxidised, Oxidation wumber of fluorine in FO is:, {a) 44 (b) +2 () -1 (4) 2, Itmthe compounds KMnO, and K2Cr2Q,, the highest, oxidation state is of the element:, , , , {a}ypotassium (b) chromium, , {c) oxygen (d) manganese, , The oxidation numbers of c in, CH,,CH,Cl,CH,Cl2,CHCl; and CCly = are, Tespectively:, , (a) 44, +2 2,-4 (b) +2, +4, 0, 4, -2, , , , , , {c) —4, -2, 0, +2, +4 (d) -2, 4, 0, +4, +2, , In which of the following reactions has the underlined, , substance been reduced?, , {a) Carbon monoxide + copper oxide —> carbon dioxide, +o, , {b)C, , , , oxide + hydrochloric acid —— > copper, , + waler, , {c) Hydrogen + iron oxide —> iron + water, , {d) Steam + iron — iron oxide + hydrogen, , In which one of the following changes, there is transfer of, , five clectrons?, , (a) Mn03 —>Mn?* (6) Cro} —> cr3+, , {c) MnO —» MnO, — (@) G07" —4 20°**, , The oxidation number of phosplhiorusiin Ba(HzPOz)2, [ur 1990), , (a) +3 (d) -1, , {b) +2 (c) #1, , 9 friendspuram extension paschimpuri sikandfa agra, ph. 9719511626
Page 2 :
New. Dimension Institute, satyaveer sir, , 27., , 29., , 3., , 32,, , $3:, , 35., , 37., , 38., , The oxidation states of the most electronegative clement in, the products of the reaction between BaO, and H,SO4, , are: [ur'1991], (a) 0 and -1 (b) -1 and -2, (c) -2 and 0 (d) -2 and +1, , In the reaction, 4Fe +30; —> 4Fe** + 607, which of the following statements is incorrest?, , (a) It is a redox reaction., , (b) Metallic iron is a reducing agent., , (©) Fe** is an oxidising agent., , (a) Metallic iron is reduced to Fe?* ., , Which of the following halide ions is easiest to oxidise?, (a) F~ (b) cm (c) Br @r, , Which of the following compounds has hydrogen with, oxidation number -1?, , (a) HCL (b) NaOH, , (c) LiH (d) HBr, , The oxidation number of chlorine in HOC! is:, (a) -1 (b) zero, , (c) +1 (d) 42, , In acid_medium)the reaction, MnO] ——» Mn?° is:, (a)/ Oxidation by 3 electrons, (b) rediiction by 3 clectrons, (c) oxidation by 5 electrons, (d) reduttion by 5 electrons, For the redox reaction,, MnO; +C,0}° +H* —> Mn?* +O, +H,0, the correct coefficients of the reactants for the balanced, , equation are: [MP PMT 2010), MnO; c,0y Ht, , (a) 2 5 16, , (b) 16 s 2, , (c) 5 16 2, , (@) 2 16 5, , The oxidation number of phosphorus in por, Py Ojo and, , POF” is:, , (a) +5 (b) 43, , (c) -3 (d) 42, , The oxidation state of Cr in KyCr 0, ist, , (a) +7 (b) +6, , (c) 43 (d) +2, , Oxidation number of *S" in NazS4Og is:, , (a) +0.5 (b) 2.5, , (c) +4 (d) +6, , Which one of the following is not a redox reaction?, (a) CaCO, —> CaO0+ CO, (b) 2H, +0; —> 22.0, (c) Na +H,O—> NaOH + 5 Hp, (4) MnCl; > MnCl, + ; cl;, Whichesubstanceis serving as a reducing agent in the, following reaction?, 14H* + CyOF- + 3Ni —> 2Cr3* +7H,0+3Ni2*, , (a) HO (b) Ni, () H* (a) Cr,07, 41., , 42., , 47,, , 49,, , 51., , Page 2 of 2, , ‘The oxidation state of iodine in H4lO% is:, , @ys7 {b) -1, , fe) +5 (d) +1, Oxidation number of N in NHyNOs is:, (a) -3 (b) +5, , (c) =3 and +5 (d) +3 and -S, , The clement, which shows minimum oxidation number in, its compounds, is:, , {a) Fe (b) Mn, , {c) Ca (d) K :, In which of the following compounds, the oxidation, number of iodine is fractional?, , (a) IF, (b) Ly, , (©) IK (d) IF,, , A compound contains atoms A, B and C. The oxidation, number of A is +2, of B is +5 and of C is -2. The possible, formula of the compound is:, (a) ABC (b) BAtAC3)2, (c) A3(BCg)2 () A3(BgC)2, For the redox reaction,, HgCl, + SnCly =~ 2Hg + SnCls, the correct coefficients oferéactants for the balanced, equation are:, (a) 1,1 (b) 1,2 (ey 2, 2 (d) 2,1, The valuc of n inthe following equation is:, , Cr Qjs, PI4H® + ne —> 2Cr3* +7H,0, (a) 2 (by 3 4 (d) 6, Why is thefollowing reaction not possible?, , G0} + Fe* +H 5.4.4..., {a) Both Cr,07> and Fe** are reducing agents, (b) Both Cr,07" and Fe** are oxidising agents, (@wCr, 07" is a strong oxidising agent while Fe* is a, weak oxidising agemt, , (d) The solution is acidic in nature, Which one of the following statements is not correct?, (a) Oxidation number of S in (NH4)7 20x is +6, (b) Oxidation number of Os in OsO, is +, (c) Oxidation number of $ in HjSOs is +8, (d) Oxidation number of O in KO, is - ;, , The oxide which cannot act as a reducing agent, is:, , (a) SO, (b) NO, , (ce) CO (d) ClO,, , Which of the following is a redox reaction?, , (a) Hy SO, and NaOH, , (b) In atmosphere, O, from O», , {c) Nitrogen oxides from nitrogen and oxygen by, lightning, , (d) Evaporation of H2O, , Number of moles of Ky Cr0) reduced by one mole of, , Sn?* ion is:, , (a) 3 (b) 3 {c) U6 (@) 6, , The metal oxide which decomposes on heating is:, , {a) ZnO : HgO, , {c) Al,O, id) Cud, , 9 friendspuram extension paschimpuri sikandfa agra, ph. 9719511626