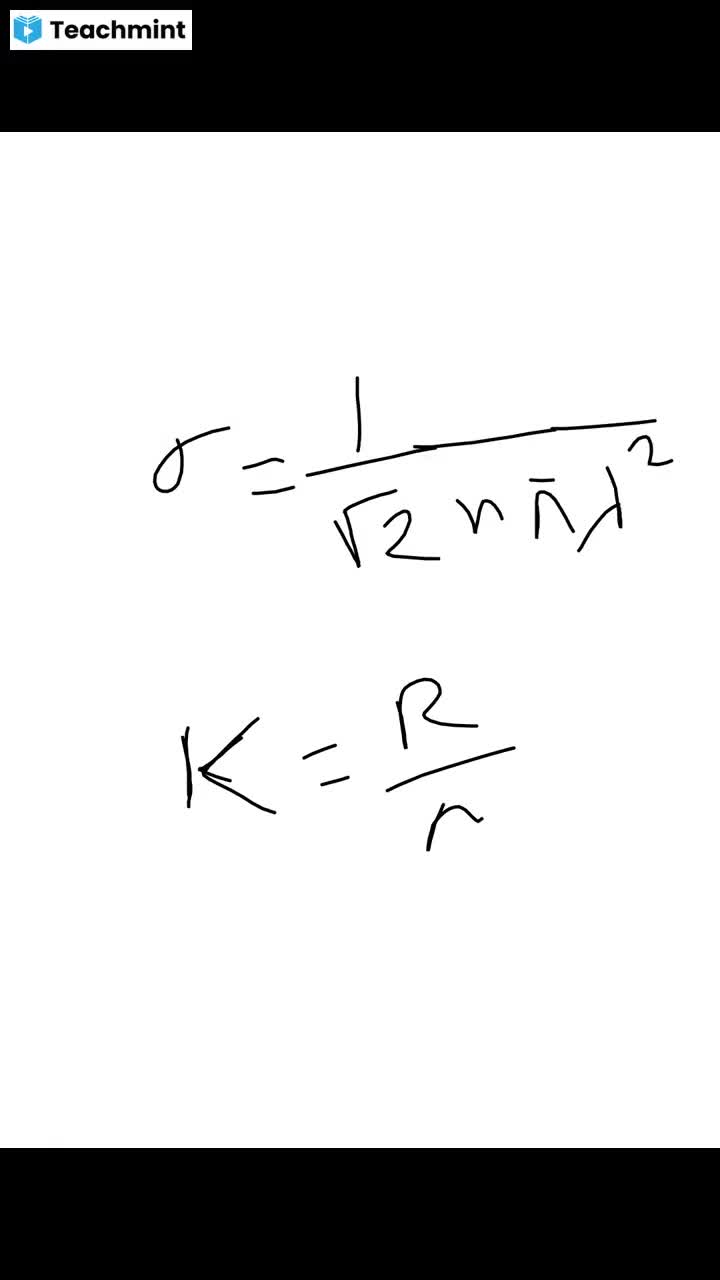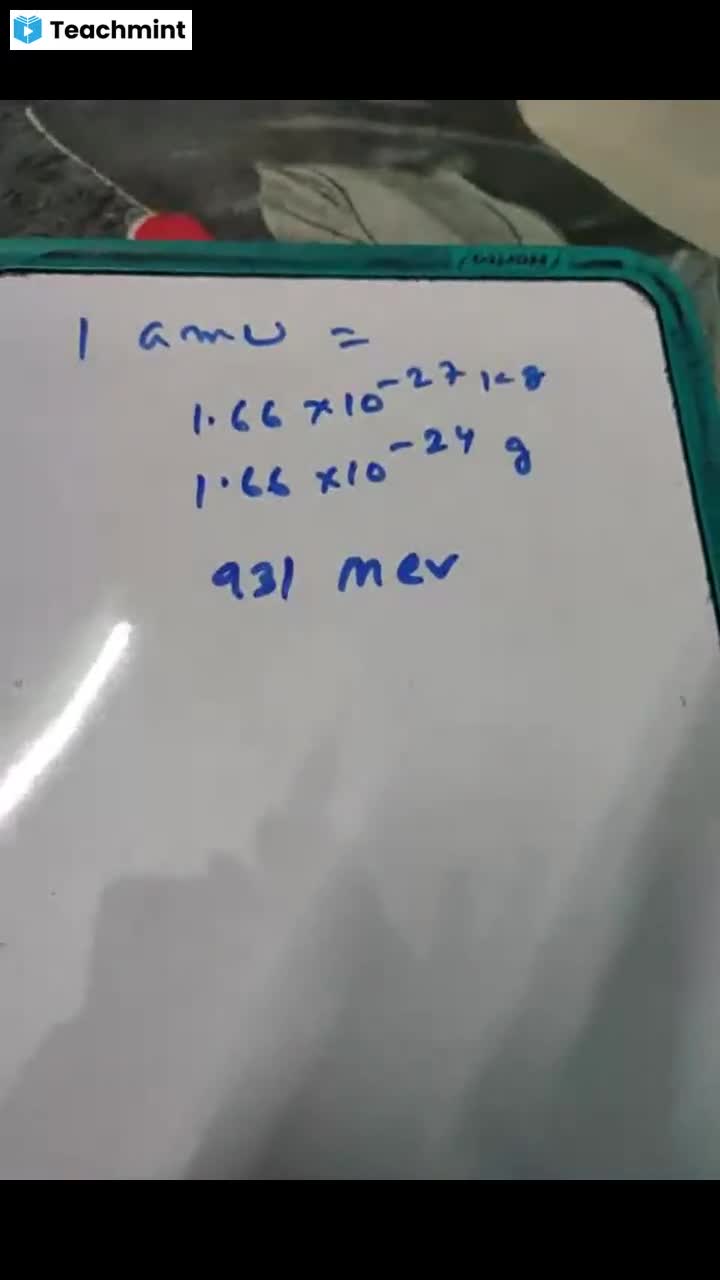Question 1 :
If atomic number is 9 and number of neutrons is 10, then find number of protons.
Question 2 :
Mass of neutron is nearly equal to that of electron. TRUE OR FALSE?
Question 3 :
A neutron is formed by an electron and a proton combining together. Therefore, it is neutral.
Question 4 :
An atom is divisible and consists of charged particles. TRUE OR FALSE?
Question 5 :
If Bromine atom is available in the form of, say, two isotopes $^{79}_{35}Br$ (49.7%) and $^{81}_{35}Br$ (50.3%), calculate the average atomic mass of bromine atom.
Question 6 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b1d2d4f59b460d7261f63a.PNG' />
With the help of the above table, find out the mass number of oxygen and sulphur atom.
Question 7 :
If atomic number is 9 and number of neutrons is 10, then find number of electrons.
Question 8 :
If number of electrons in an atom is 8 and number of protons is also 8, then what is the charge on the atom?
Question 10 :
The mass of an electron is about $\frac{1}{2000}$ times that of proton.
Question 12 :
What is the SI prefix for $10^{-12}$ multiples and submultiples of a unit?
Question 20 :
Compute the ratio by mass of the combining elements for Carbon monoxide.
Question 23 :
The visible universe is estimated to contain $10^{22}$ stars. How many moles of stars are present in the visible universe?
Question 28 :
Hydrogen and oxygen combine in the ratio of 1 : 8 by mass to form water. What mass of oxygen gas would be required to react completely with 3 g of hydrogen gas?
Question 30 :
A change in the physical state can be brought about ___.
Question 31 :
How many electrons are there in the L shell of chlorine atom ?
Question 33 :
Rutherford’s α-particle scattering experiment led to the discovery of the ____________.
Question 35 :
Which of the following is the correct electron distribution of chlorine atom ? (Atomic number of chlorine is 17).
Question 37 :
Atomic models have been improved over the years. Arrange the following atomic models in the order of their chronological order
(i) Rutherford’s atomic model
(ii) Thomson’s atomic model
(iii) Bohr’s atomic model
Question 38 :
Which of the following scientist identified the 'Indivisibility of atoms' in the structure of an atom ?
Question 40 :
The ratio of the radii of hydrogen atom and its nucleus is ~ $10^{5}$. Assuming the atom and the nucleus to be spherical and if atom is represented by planet earth $‘R’=6.4\times 10^{8}m$ , estimate the size of the nucleus.
Question 41 :
An atom with 3 protons and 4 neutrons will have a valency of
Question 42 :
Which of the following scientist identified the 'Canal rays' in the structure of an atom ?
Question 44 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b1d2b1f59b460d7261f60b.JPG' />
Find out the valency of atoms represented by the figure.
Question 45 :
Which of the following statements about Rutherford’s model of atom are correct?
(i) considered the nucleus as positively charged
(ii) established that the $\alpha$–particles are four times as heavy as a hydrogen atom
(iii) can be compared to solar system
(iv) was in agreement with Thomson’s model
Question 46 :
Which of the following scientist identified the 'Neutron' in the structure of an atom ?
Question 47 :
The number of electrons in an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of the element?
Question 48 :
Dalton’s atomic theory successfully explained:
(i) Law of conservation of mass
(ii) Law of constant composition
(iii) Law of radioactivity
(iv) Law of multiple proportion
Question 50 :
<img style='object-fit:contain' src='https://teachmint.storage.googleapis.com/question_assets/cbse_ncert/61b1d2b1f59b460d7261f60a.JPG' />
Which of the above in the figures do not represent Bohr's model of an atom correctly?