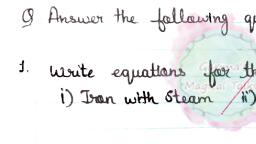Page 1 :
Class 8 Chemistry Metals and Non metals, , Introduction, , Introduction, In our day to day life we use many well-known materials like iron, aluminum, copper, gold, silver and many more. We are also familiar with the elements, like oxygen, carbon, Sulphur as well. These are all obtained from some naturally occurring substances encompassing one or more elements or their, compounds. They are termed as minerals. Minerals form which elements can be profitably extracted are termed as ores., , Fig. Ores and Minerals, , There 92 well known naturally occurring minerals of which 70 are metals and rets 20 are the non-metals., Some metals possess the characteristics of both metals as well as non-metals. They are termed as metalloids. For instance, boron, silicon,, germanium, arsenic, antimony, tellurium, and polonium., Elements found in free state includes some metals like gold, silver, platinum etc. and some non-metals like helium, neon, argon etc. as well., A major proportion of metals are found in combined states like oxides, sulphides, carbonates, silicates etc. Some non-metals like sulphur,, phosphorous too are found to exist in combined state.
Page 2 :
Class 8 Chemistry Metals and Non metals, , Physical properties of metal, , Physical properties of metal, Metals are solid except mercury that is found in liquid state at room temperature., , They are hard and tough except sodium and potassium that can be cut with a knife., They are lustrous i.e. they shine in light because metals possess free electrons that vibrates on getting in contact with light., , Fig. The key made of metal shines in light that implies metal is lustrous, They have high melting points except caesium and gallium have very low melting point., They are good conductors of heat and electricity. The best conductors of electricity are silver and copper whereas lead and mercury are, comparatively poor conductors., , Fig. Metal is used at the tip of the plug that is inserted into the socket, They have high density except alkali metals like lithium, sodium and potassium., They are malleablee. they can be beaten into thin sheets. For instance, the aluminium foil we use to pack food is manufactured by beating the, aluminium metal into thin sheets. This a characteristic property of metal.
Page 3 :
Fig. Aluminium is beaten into thin sheets to make aluminium foil used to pack food, They are ductilee. they can be drawn into wires. For instance. We all are familiar with copper and aluminium wires. It is due to their characteristic, property of ductility that these metals can be drawn into wires., , Fig. copper aluminium are drawn into wires, They are sonorouse. they produce a ringing sound when struck against a hard object. For instance, your school bells are made of metal that, produces a ringing sound after the period is over when struck hard by a hammer., , Fig. bells in temples are made of metal that when struck against hard surface produces sound, Examples are iron, gold, silver, magnesium, copper, aluminium and many more
Page 4 :
Class 8 Chemistry Metals and Non metals, , Physical properties of non-metals, , Physical properties of non-metals, Non-metals may be either solids, liquids or gases., Solid non-metals are brittle and break down into powdery mass on striking with a hammer except diamond which is the hardest non-metal., They have a dull luster but iodine is lustorous., They have low melting points except diamond that has very high melting point., They are poor conductors of heat and electricity except graphite., They are not malleable., They are not ductile., They are not sonorous., Examples are carbon, Sulphur, oxygen, phosphorous and many more., , Fig. Bonding of non-metallic elements
Page 5 :
Class 8 Chemistry Metals and Non metals, , Reactions of metal with oxygen, , Reactions of metal with oxygen, Metals reacting with oxygen is observed in our day to day life. We all must have observed rusting of iron, silver jewellery getting tarnished, or copper, articles getting covered by green layer. Metals reacts with atmospheric oxygen and produces metal oxides that are basic in nature because they react, with water to form bases., In case of rusting of iron, the iron reacts with the oxygen present in air and moisture and develops rust (hydrated iron (III) oxide)., , Fig. Iron chain left in moist air got rusted, In case of rusting of copper, the metallic copper reacts with oxygen, carbon-dioxide and atmospheric moisture and develops a green coloured, coating of copper hydroxide and copper carbonate., , Fig. Copper developing green coloured rust on exposure to moist air, In case of tarnishing of silver articles, the metallic silver reacts with hydrogen sulphide or sulphur present in air and gets tarnished., , Copper burns in air to combines with oxygen and form copper (II) oxide, a black oxide.
Page 6 :
Class 8 Chemistry Metals and Non metals, , Reactions of metal with water, , Reactions of metal with water, Sodium being very reactive reacts vigorously with water leading to the production of sodium hydroxide and hydrogen. Therefore it is stored in, kerosene., , Magnesium undergoes reaction with water to form magnesium hydroxide and hydrogen., , Whereas iron reacts very slowly with water.
Page 7 :
Class 8 Chemistry Metals and Non metals, , Reactions of metal with acids, , Reactions of metal with acids, Metals react with acids to produce hydrogen gas. If a matchstick is brought near the mouth of the tube containing the product of the reaction then we, hear a pop sound. It is this hydrogen gas that burns with a pop sound., For instance, Magnesium reacts with dilute hydrochloric acid to form magnesium chloride and hydrogen., , Copper does not react with hydrochloric acid but reacts with sulphuric acid.
Page 8 :
Class 8 Chemistry Metals and Non metals, Reactions of metal with bases, Metals react with sodium hydroxide to produce hydrogen gas., For instance, zinc reacts with sodium hydroxide to give sodium zincate., Zn + 2NaOH -> Na2ZnO2 + H2, , Reactions of metal with bases
Page 9 :
Class 8 Chemistry Metals and Non metals, , Reactions of non-metal with oxygen, , Reactions of non-metal with oxygen, Non-metals reacts with oxygen to produce non-metallic oxides that are acidic in nature because when dissolved in water non-metallic oxides gives off, non-metallic acids., Sulphur reacts with oxygen producing sulphur dioxide which on further reaction with water gives sulphurous acid., S + O2 -> SO2, SO 2 + H2O -> H2SO 3
Page 10 :
Class 8 Chemistry Metals and Non metals, , Reactions of non-metal with water, , Reactions of non-metal with water, Non-metals do not react with water but are very reactive in air due to which they are stored in water. Phosphorous is a vigorously reacting non-metal, that catches fire if exposed to air. To avoid such reaction phosphorous is stored in water that cuts off its contact with atmospheric oxygen.
Page 11 :
Class 8 Chemistry Metals and Non metals, , Reactions of non-metal with acids, , Reactions of non-metal with acids, Non-metals do not react with acids. This is due to the fact that a substance undergoing reaction with acids, donates electrons to the H+ ions produced, by the acids. But non-metals are acceptor of electrons and hence they do not donate electron to the hydrogen ion of the acid. As a result they do not, react with dilute acids.But there are some exceptions., 6HNO 3 + S -> H2SO 4 + 6NO2 + 2H2O
Page 12 :
Class 8 Chemistry Metals and Non metals, Reactions of non-metal with bases, Reactions of non-metals with bases are quite complex. They react with bases to form salt., Reaction of calcium hydroxide and chlorines gives bleaching powder., Ca(OH)2 + Cl2 -> Ca(OCl)Cl + H2O, , Reactions of non-metal with bases
Page 13 :
Class 8 Chemistry Metals and Non metals, , Displacement reaction in metals, , Displacement reaction in metals, In displacement reaction a more reactive metal displaces a less reactive metal but a less rwactive metal cannot displace a more reactive metal., , Let us take 5 beakers., , In beaker A Zn being more reactive than copper replaces copper from the solution of copper sulphate and forms zinc sulphate., CuSO4 + Zn --> ZnSO4 + Cu, In beaker B iron being more reactive than copper replaces it from its solution and forms iron sulphate., CuSO4 + Fe --> FeSO4 + Cu, In beaker C there would be no reaction as less reactive metal (copper) cannot displace more reactive metal (zinc) from its solution., In beaker D there would be no reaction as less reactive metal (copper) cannot displace more reactive metal (iron) from its solution., In beaker E there would be no reaction as less reactive metal (iron) cannot displace more reactive metal (zinc) from its solution.
Page 14 :
Class 8 Chemistry Metals and Non metals, , Uses of metals, , Uses of metals, Metals being hard, tough are used in manufacturing automobiles, machinery, trains, satellites, aeroplanes, industrial gadgets., , Due to its characteristic of being a good conductor of electricity and ductility it is used in making wires, electrical appliances, circuits and many, more., , Being a good conductor of heat it is used in making utensils, water boilers.
Page 15 :
Class 8 Chemistry Metals and Non metals, Uses of non-metals, Non-metal is the basic requirement of life. Oxygen is inhaled while breathing., Nitrogen is used in fertilizers to enhance the fertility of soil., Chlorine and fluorine are used for water purification., It is used as an antiseptic and applied on wounds., It is used as disinfectant., It is used in crackers., , Uses of non-metals