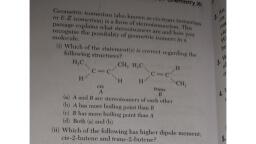Page 1 :
GEOMETRICAL ISOMERISM (RESTRICTED ROTATION), When the same molecular formula represents two compounds which differ in the spatial, arrangement of atoms or groups around carbon-carbon double bond, then such isomers are called, geometrical isomers. The phenomenon is called geometrical isomerism. Geometrical isomerism is, due to the restricted or hindered rotation around the double bond. Geometrical isomers are also, called Diastereomers. In case of organic compounds, the rotation around (C - C) single bonds is free, but the rotation around (C = C) double bonds is restricted. The two carbons of the carbon-carbon, double bond are sp2 hybridized. The three sp2 hybrid orbitals lie in one plane. Thus, the three sigma, bonds formed by each of these two carbon atoms are co-planar. Each carbon atom is left with one, unhybridized p-orbital at right angles to the sp2 hybrid orbitals. These two p-orbitals overlap to form, the Pi-molecular orbital. The Pi-cloud spreads over both carbon atoms above and below the plane of, sigma bond (C - C). Thus, a (C = C) double bond consists of one 𝜎-bond and one 𝜋-bond., The overlap of two unhybridized p-orbitals is maximum when they are parallel to each other. This is, possible only if all the four groups (attached to the two carbon atoms) alongwith the two carbon, atoms are co-planar as shown in Fig. (1)., , X, , X, C, , , X, , C, , , , Y, C, , Y, , Y, , , , , , Y, , C, X, , , , Cis, , Trans, , Geometrical isomers Fig.1, Hence, any change in this co-planar geometry by twisting the (C = C) bond would make the molecule, unstable. Therefore, rotation around carbon-carbon double bond would be restricted., , On account of the restricted or hindered rotation about the double bond, two different spatial, arrangements are possible. These cannot be interconverted easily. For example, the molecular, formula, C2 H2Cl2 can represent two structures., H, , Cl, , Cl, , C, C, Cl, , H, , cis, , H, C, C, Cl, , H, , trans, , The the above two structures will be exactly identical if there were rotation about carbon-carbon, double bond. Due to the restricted rotation around C = C, the above structures represent two, distinct compounds. The isomer in which two similar atoms are on the same side of the double bond, is known as cis- isomer and the other in which the two similar groups are present on the opposite, side of the double bond is known as trans isomer. This type of isomerism is known as Geometrical or, cis-trans isomerism., Conditions for Geometricai 1somerism, (i) 'The molecule must have a carbon to carbon double bond, ii)There must be two different atoms or groups attached to each carbon atom of the double bond. if, one or both the carbons of the double bond carry two similar atoms or groups, then no geometrical, isomerism is possible. for example, propene and isobutene do not slow geometrical isomerism.
Page 2 :
CH3, , H, C, , C, , C, , H, , H, , H3C, C, , H3C, , H, , H, , isobutene, , Propene, , Charncteristies of Geometrieal isonmers., (i) Gieometnic isomers have similar chemical properties. ( They have dilferent physical properties, sach as melting points, dipole moments, refractive index, and adsorption capacity on materials used, in chromatographic analysis., The difference in the physical properties of geometrical isomers can be used for the assignment of, cis and trans- configurations to a compound, "The cis-isomer, generally, has a lower melting point, than the corresponding trans-isomer, whereas the boiling point, solubility and refractive index of the, cis isomer are higher than those of the corresponding trams isomer., The cis-isomers show a large dipole moment due to their unsymmetrical structure while trans, isomers have zero or negligible dipole moment due to their symmetrical structure. The individual, bond moments in case of trans-isomers cancel each other because they act in opposite directions., The higher values of the dipole moment in case of cis isomers means higher polarity which is, responsible for the higher boiling points of cis isomers. The trans isomers due to their symmetrical, structure fit well into crystal lattices. Consequently, trans- isomers have, generally, higher melting, points than the cis- isomers (due to their poor symetry)., Determination of Configuration of a geometrical isomer. Following physical propertics, are used to, determine the configuration of the geometrical isomer., (i)From dipole moment. Cis-Isomer has a higher value of dipole moment as compared to its transisomer.In Cis-isomer, the similar groups are on the same side and the electronic moments or pole, moments are additive, But in case of trans isomer, the pole-moments being on opposite sides, the, net dipole moment is either zero or substantially decrease., H, , H3C, , H, , H 3C, C, , C, , H, , C, , C, H, , CH3, , Trans 2-Butene, , =0, , H 3C, , cis-2-Butene, , = 0.4, , Cl, , Cl, , C, , H, C, , H, C, H, , C, Cl, , H, , trans, cis = 0.4, 1,2-Dichloroethene, , Cl, , =0, , (ii) From melting point. The melting point depends upon the symmetry of the molecule. A trans, isomer is more symmetrical and can fit more closely in its crystal lattice. Due to this, trans isomer, has higher melting point than cis- isomer. In cis-isomer, the two bigger groups on the same side, cannot fit closely in the lattice. e.g. Maleic acid (cis) isomer melts at 403 K whereas Fumeric acid, (trans) melts at 575 K., (iii) From boiling point. Cis-isomer has higher value of dipole-moment and is more polar as compared, to its trans-isomer. The higher polarity binds the molecule, more strongly in cis-isomer and hence it, boils at a higher temperature. For example : cis-2-butene boils at 277 K whereas trans-2- butene