Page 1 :
Carbohydrates
Page 2 :
Carbohydrates are a class of naturally occurring compounds and, derivatives formed from them., In the early part of the 19th century, substances such as wood, starch, and linen were found to be composed mainly of molecules containing, atoms of carbon(C), hydrogen (H), and oxygen (O) and to have the, general formula C6H12O6;, Organic molecules with similar formulae were found to have a similar, ratio of hydrogen to oxygen., The general formula Cx(H2O)y is commonly used to represent many, carbohydrates, which means “watered carbon’’
Page 3 :
Definition of Carbohydrates, Carbohydrates are defined as “polyhydroxy aldehydes or, ketones or the compounds which yield these on hydrolysis”.
Page 5 :
Sugars, , Non-Sugars, , They are crystalline in They are non, nature, crystalline in nature, Soluble in water, , Insoluble in water, , Sweet in taste, , Tasteless
Page 10 :
sugars, , Reducing, Sugars, , Non-Reducing, Sugars
Page 11 :
Reducing Sugars, , Non-Reducing Sugars, , Sugars which reduce, Fehling’s solution or, tollen’s reagent are called, reducing sugars, Examples, Glucose, Galactose,, Maltose, Lactose, , Sugars which do not reduce, Fehling’s solution or, tollen’s reagent are called, non-reducing sugars, Examples, Sucrose, Trehalose
Page 12 :
Haworth structures or Haworth Projections, The cyclic forms of sugars are written as hexagons and, pentagons. These are called Haworth projections. The, cyclic forms of sugars are convenient to draw., The formation of Intramolecular hemiacetal or hemiketal, is the basis for glycosidic formation in carbohydrates., Intramolecular hemiacetal formation results in a ring form, which is either a five membered ring with oxygen atom or, a six membered ring with one oxygen atom.
Page 18 :
Glucose, CHO, H, OH, HO, H, H, OH, OH, H, CH 2 OH, , H, H, HO, H, H, , OH, OH, O, H, OH, CH2OH, , D-Glucose, , α-D-Glucose, , Open chain structure, , Cyclic structure
Page 19 :
Glucose, H, H, HO, H, H, , OH, OH, O, H, OH, CH2OH, , CH 2OH, OH, H, H, OH H, HO, OH, H, OH, , α-D-Glucose, , α-D-Glucopyranose, , Cyclic structure, , Haworth structure
Page 20 :
Galactose, , CHO, H, OH, HO, H, HO, H, OH, H, CH 2 OH, , H, H, HO, HO, H, , OH, OH, H O, H, CH2OH
Page 22 :
Mannose, CHO, HO, H, HO, H, H, OH, OH, H, CH 2OH, , H, HO, HO, H, H, , OH, H, H O, OH, CH2OH
Page 24 :
Glucose,Galactose and mannose are reducing sugars., This is because they have an alpha hydroxy aldehyde, function and gives a hemiacetal structure in, equilibrium with the open chain form having a free, aldehyde group., Such sugars therefore reduce Fehling’s solution and, tollen’s reagent., , Reducing sugars also exhibit mutarotation.
Page 25 :
Fructose, CH 2OH, O, HO, H, H, OH, OH, H, CH 2OH, D-Fructose, Open chain form, , HOH 2C, HO, H, H, , OH, H, OH, , O, , CH 2OH, α-D-Fructose, Cyclic form
Page 27 :
Fructose, HO, HO, H, H, , CH 2OH, H, O, OH, CH 2OH, , β-D-Fructose, Cyclic structure, , HO, , O OH, H HO, H, OH H, , OH, , β-D-Fructofuranose, Furanose Structure
Page 28 :
Fructose, , HOH 2C, HO, H, H, H, , OH, H O, OH, OH, H, , α -D-Fructose, Cyclic structure, , H, , OH, O, H, H H OH, HO, OH, OH H, α -D-Fructopyranose, , Pyranose Structure
Page 29 :
HO, HO, H, H, H, , CH 2OH, H O, OH, OH, , OR, , HO, HO, O H, H, , CH2OH, H, OH, OH, H, H, , H, , H, H, , O OH, H H OH, , HO, OH, , H, , OH, , β-D-Fructose, , β-D-Fructopyranose, , Cyclic structure, , Pyranose Structure
Page 30 :
Fructose is a reducing sugar., This is because it has a alpha hydroxy ketone function, and gives a hemiketal structure in equilibrium with the, open chain form having a free keto group., Therefore fructose reduces Fehling’s solution and, tollen’s reagent., , Reducing sugars also exhibit mutarotation.
Page 31 :
Sucrose or Saccharose, (Table sugar, Cane Sugar, Beet sugar, House hold, sugar), It is also called invert sugar as on hydrolysis it yields, equimolar mixture of glucose and fructose, Systematic name :α-D-glucopyranosyl-(1 –>2) –β -Dfructofuranose, It is composed of equimolar amounts of α-D-glucose, and β -D-fructose.
Page 32 :
Haworth structure, OH, H, , OH, , H, OH, , H, , H, , OH, , HO, , 0, , HO, O, H, HO, H, OH, , H, , OH
Page 33 :
Sucrose is a non reducing sugar. This is because the, anomeric carbons of both monosaccharides are, involved in glycosidic or acetal formation., The hemiacetal structure is not present that can be, opened up to give a free aldehydic group., Hence it does not reduce Tollen’s reagent or, Fehling’s solution., , It does not exhibit mutarotation.
Page 34 :
Maltose, Systematic name : 4-0-α-D-glucopyranosyl-Dglucopyranose, It is a disaccharide obtained from partial, hydrolysis of starch., It consists of two α-D-glucose residues which are, condensed together through glycosidic linkage. α1,4 glycosidic linkage)., Maltose is present in germinating seeds, particularly cereal. It does not occur abundantly in, nature.
Page 35 :
Maltose, Haworth structure, OH, , OH, H, , H, OH H, , OH, , HO, H, , OH, , H, 0, , H, OH H, , OH, OH, , H, , OH
Page 36 :
Maltose is a reducing sugar, This is because in one of the monosaccharide, units there is a hemiacetal group that can be, opened up to give a free aldehydic function.
Page 38 :
Lactose, Haworth structure, OH, , OH, O, OH, H, OH H, H, H, H, OH, , H, 0, , H, OH H, , OH, OH, , H, , OH
Page 39 :
Lactose is a reducing sugar., This is because in one of the monosaccharide, units there is a hemiacetal group that can be, opened up to give a free aldehydic function.
Page 40 :
Epimerisation, The change of configuration of one of the asymmetric, carbon atoms in compounds containing two or more, asymmetric carbon atoms is known as Epimerisation., Epimers are aldoses which produce the same osazones., , They have the same configuration on all their, asymmetric carbon atoms except the alpha carbon.
Page 42 :
Epimers, Aldoses which have identical configuration on, all their asymmetric carbon atoms except alpha, carbon atom and produce the same osazone are, known as Epimers
Page 50 :
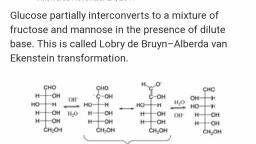
Osazone formation involves only the first two carbon, atoms in a sugar. It therefore follows that the, configuration of the rest of the molecule in glucose and, Fructose must be the same. Hence D-glucose gives the, same osazone as Fructose natural D(-)-fructose.
Page 53 :
Conformations of α-D-glucose and β-D-glucose, The pyranose ring of glucose is not flat., Hence Haworth structures are inadequate in representing, carbohydrate conformations., Hence the best representation of cyclic structures of glucose, would be in terms of their conformational structure., , X-ray crystallographic studies and other physical measurements, indicate that the pyranose ring of glucose preferentially adopts a, chair conformation.
Page 54 :
OH, , H, HO, HO, , H, H, , H, , OH, , H, , O, H, , OH, HO, , α-D-glucose, , HO, HO, , H, H, , H, , O, OH, OH H, , β-D-glucose
Page 55 :
Chair conformation is the most stable conformation, that a six membered ring can exist in., β -D-glucose form of glucose is more stable than αD-glucose form of glucose., , This is because in β form, all bulky groups are in, equatorial positions which involves lesser crowding, between the bulky groups., Stability, β-D-glucose > α-D-glucose
Page 56 :
Mutarotation, Mutarotation was discovered by French chemist Dubrunfaut in, 1844, when he noticed that the specific rotation of aqueous sugar, solution changes with time, The phenomenon of spontaneous change of specific rotation of, an optically active compound, with time, to an equilibrium value, is called mutarotation.
Page 57 :
D-glucose exists in two crystalline forms, α– D- glucose and β -D-glucose, When either forms of D-glucose is dissolved in water, and allowed to stand a gradual change in specific, rotation occurs., , The specific rotation of the alpha form falls and that, of the beta form rises until a constant value of, +52.70 (530) is reached.
Page 58 :
Explanation, Glucose forms a stable cyclic hemiacetal between the CHO group and the -OH group on the fifth carbon., As a result of this the first carbon becomes, asymmetric giving two isomers I and I which differ, only in the configuration of the asymmetric carbon.
Page 59 :
The carbon atom of the functional group (-CHO, or C=O ) after cyclisation becomes chiral and are, called Anomers., , Thus I and II are called anomers., The anomer I with -OH group to the right is, designated as alpha-D-glucose and the, anomer II with -oH group to the left is, designated as beta-D-glucose.
Page 60 :
H, H, HO, H, H, , OH, OH, H O, OH, CH 2OH, , α-D-glucose, , H, HO, H, H, , CHO, OH, H, OH, OH, CH2OH, , Open chain form, , HO, H, HO, H, H, , H, OH, H O, OH, CH 2OH, , β -D-glucose
Page 61 :
Mutarotation occurs because of the slow, interconversion of α-D-glucose and β-D-glucose via the, open chain form until equilibrium is established giving, a constant specific rotation of +530., The phenomenon of mutarotation can be explained on, the basis of the cyclic forms of glucose. The initial, rotations of +1120 and +190 are caused by the presence, of α-D-glucose (+1120) or (+190) respectively. In the, solid state these two forms of glucose are stable.
Page 62 :
The ring formation from open chain structure to form, α -D-glucose and β -D-glucose is due to the formation, of hemiacetal between aldehyde group and one, alcoholic group of the same molecule., , The isomeric forms α -D-glucose and β -D-glucose are, called anomers and this phenomenon is called, anomeric effect., The ring formation is assumed as a typical hemiacetal, formation in which an alcoholic group of the sugar is, involved with the aldehyde group. The ring form is, called the lactol form.
Page 63 :
OH, H, , H, OH H, , OH, , HO, , OH, H, , OH, , α-D-glucose, I, , H, HO, H, H, , CHO, OH, H, OH, OH, CH 2OH, , OH, H, , H, OH H, , O OH, , HO, , H, H, , OH, , D-Glucose, β-D-Glucose, Linear form, II, Mutarotation Equilibrium
Page 64 :
Mechanism, Mutarotation involves a simultaneous attack by, an acid and a base to produce the open chain, aldehyde structure., Therefore water is an amphiprotic solvent acting, as an acid and as a base., Thus water acts as proton acceptor becoming, H3+O and also proton donor becoming OHAll reducing sugars(except a few ketoses) exhibit, mutarotation
Page 65 :
The phenomenon of mutarotation occurs slowly in, aqueous medium., However its rate can be enhanced either by heating, the solution or by adding some Catalyst such as an, acid or base., when either form is placed in solution it slowly forms, the other via the open chain aldehyde form and the, gradual change in specific rotation is attributed to, the establishment of equilibrium between the two, forms., , The final value +530 corresponds to the equilibrium, mixture of the α and β forms.
Page 66 :
OH, H, , +, , H ----OH, -, , H, OH H, , HO, , O-H, H, , OH, , α-D-glucose, I, , -, , H, , OH, H2O, , +, , OH, , H, , OH, , -, , H, OH H, , OH, , HO, OH, , OH, H, , O, H, , -, , H3O, , +, , H, OH H, , +, , H ----OH, , OO - H, , HO, , H, H, , OH, , H2O, , D-Glucose, β-D-Glucose, Linear form, II, Mutarotation Equilibrium, , -
Page 67 :
I, , II
Page 68 :
Representation of structures of monosaccharides, (DL notation), Monosaccharide with the same configuration on the highest, numbered chiral carbon as D (+) glyceraldehyde is designated, as a D-monosaccharide, A monosccharide in which the highest numbered chiral, carbon has the configuration as L-glyceraldehyde is, designated as belonging to L-series, D and L designations have no particular relation to the signs, (+) or (-)
Page 69 :
1, , CHO, H, OH, HO, H, H 4 OH, H 5 OH, CH 2OH, 2, 3, , 1, , CHO, H, OH, HO 3 H, H 4 OH, 5, HO, H, CH 2OH, 2
Page 70 :
H, HO, H, H, , CHO, OH, H, OH, OH, CH 2OH, , D-glucose, , H, HO, H, HO, , CHO, OH, H, OH, H, CH 2OH, , L-glucose
Page 71 :
Disadvantage of DL notation, DL system of stereochemical designations has the, disadvantage of specifying the configuration at one chiral, centre., But still D-L system of stereochemical designations is still used, in carbohydrate literature to indicate the designations of, carbohydrates.
Page 72 :
Open chain structure of glucose, 1)Molecular formula, Elemental analysis and molecular weight determination, show that the molecular formula of glucose is C6H12O6, 2)Presence of five -OH groups, When glucose is treated with acetic anhydride in the, presence of pyridine, glucose penta acetate is formed, showing the presence of five hydroxyl groups., C6H7O(OH)5 +5(CH3CO)2O, , C6H7O(OCOCH3)5 + 5CH3COOH
Page 73 :
As glucose is not easily dehydrated, each hydroxyl group, must be attached to a different carbon atom, This is because organic compounds with two hydroxyl, groups attached to a single carbon atom usually lose water, to produce a carbonyl group, 3) Presence of a carbonyl group, , a)Glucose reacts with HCN forming Glucose cyanohydrin, (C5H12O5)C=O + HCN, , C5H12O5 (OH) CN
Page 75 :
4) Presence of aldehyde and primary alcohol groups, When glucose is reduced with bromine water, gluconic, acid, a monocarboxylic acid with the same number of, carbon atoms is obtained., (C5H11O5)CHO, , Br2/ H2O, , (C5H11O5 )CQOH, , This reaction shows that the carbonyl group is aldehyde, because only the aldehyde group can be oxidised to an acid, having the same number of carbon atoms., Aldehyde group being a monovalent group must be present, at the end of the straight chain
Page 81 :
Emil Fischer showed that naturally occurring glucose, has the structure shown below, , H, HO, H, H, , CHO, OH, H, OH, OH, CH 2OH
Page 82 :
Demerits of Open chain structure of Glucose, , H, HO, H, H, , CHO, OH, H, OH, OH, CH 2 OH
Page 83 :
Cyclic or Ring structure of Glucose, Demerits of Open chain structure of Glucose, 1) D-(+)-glucose fails to undergo some of the typical, reactions of aldehydes:, , a)Glucose does not form a bisuphite addition product, with NaHSO3, b)Glucose does not form an addition product with NH3
Page 84 :
c) Glucose forms two isomeric pentamethyl and, pentaacetyl derivatives. Neither of these derivatives, reduce Fehling’s solution ie they are non reducing., These derivatives do not react with hydrogen, cyanide, Phenyl hydrazine and hydroxylamine, which reveals that glucose does not contain an, aldehydic group., d)Glucose does not restore the colour of Schiff ‘s, reagent.
Page 85 :
2)D-(+)-glucose exists in two stereoisomeric forms which, undergo mutarotation. The existence of these two forms is, not explained by open chain formula., 3) Formation of two isomeric methyl glucosides which do, not undergo mutarotation., Normal aldehydes when refluxed with alcohol in the, presence of hydrogen chloride consumes two molecules, of alcohol to form acetals.
Page 86 :
But glucose (although an aldehyde in open chain structure, consumes one molecule of an alcohol in the presence of dry, hydrogen chloride gas to form a mixture of two crystalline, isomeric methyl glucosides (alpha-methyl-D-glucose and, beta-methyl-D-glucoside., These methyl glucosides do not undergo mutarotation and, do not reduce Fehling’s solution or tollen’s reagent.
Page 87 :
4) when methyl-D-glucoside is treated with methyl iodide in the, presence of dry silver oxide Catalyst, complete methylation, takes place to obtain Glucose penta methyl derivative., Glucose penta methyl derivative obtained on acid hydrolysis, under mild condition gives a tetramethyl derivative which is, reducing ie it reduces Fehling’s solution., Therefore one of the hydroxyl groups in glucose has a different, reactivity from the other four hydroxyl groups., But according to the open chain structure, all the hydroxyl, groups are of the same character.
Page 88 :
5) X-ray analysis proves definitely the existence of the, ring structure and at the same time indicates the size of, the ring., To explain the above facts, a cyclic structure for glucose, was proposed. The structure of α-D-glucose is represented, as follows., , H, H, HO, H, H, , OH, OH, H O, OH, CH2OH
Page 89 :
Ring structure of glucose- Justification of cyclic structure, The above objections can be explained on the basis that glucose, does not contain a free aldehyde group., It is assumed that the aldehyde group of glucose reacts with OH group in the chain to give an internal, cyclic, hemiacetal, ring formation between C1 and C5., A new chiral carbon is generated at the carbonyl carbon of, the open chain structure, which exists in two configurations, during ring formation., That is the spatial arrangement of H and -OH has given rise to, two additional structures., , That is these two forms have been confirmed by x-ray analysis.
Page 90 :
H, H, HO, H, H, , OH, OH, H O, OH, CH 2OH, , α-D-glucose, , H, HO, H, H, , CHO, OH, H, OH, OH, CH2OH, , Open chain form, , HO, H, HO, H, H, , H, OH, H O, OH, CH 2OH, β -D-glucose
Page 91 :
The cyclic structure can account for the following observations:, a) The glucose ring is not very stable. Strong reagents like, HCN, NH2OH, NH2NHC6H5 break the ring to give the, intermediate aldehyde form., b)However weak reagents like NH3 and NaHSO3 are not able to, open the chain and cannot react with it. This explains the, inability of glucose to form aldehyde-ammonia and, bisulphite addition compounds.
Page 92 :
d)Similar indication of the cyclic nature of glucose from the, observation of another phenomenon known as mutarotation. This, phenomenon appears due to slow interconversion of alpha form, and beta form via the open chain form until equilibrium giving a, constant specific rotation of +530, According to this the two forms of glucose d and l attain an, equilibrium value of optical rotation when dissolved separately in, water even though they have different initial values. The, explanation of mutarotation that the sugar exists in solution as a, mixture of two ring forms in equilibrium with a comparitively, small proportion of glucose in the open chain or aldehyde form.
Page 93 :
c) The cyclic structure also explains that methyl glucosides do, not undergo mutarotation. The methoxy group attached to the, anomeric carbon will not open up to give a free aldehyde., Hence does not undergo mutarotation., H, H, HO, H, H, , OCH 3, OH, H O, OH, CH 2OH, , methyl α-D-glucopyranoside, (acetal)
Page 94 :
The cyclic structure of Glucose can be represented as shown below, , H, H, HO, H, H, , OH, OH, H O, OH, CH2OH
Page 95 :
Conformational structures, Sucrose(α-D-glucopyranosyl-β-D-fructofuranose), Sucrose is called table sugar or beet sugar.It is a non reducing, sugar. The glycosidic linkage is α,β-(1,1)., OH, , H, HO, HO, , H, H, , H, , O, H, OH, 0, , HO, O, H HO, H, OH, , H, , OH
Page 96 :
Maltose (α-D-glucopyranosyl-α-D-glucopyranose), , Maltose is called malt sugar. It does not occur in free state.It is, obtained by the partial hydrolysis of starch in the presence of, enzyme diastase.It is a reducing sugar. The glycosidic linkage, is α-(1,4)., , OH, , H, HO, HO, , H, H, , H, , OH, , H, O, , HO, HO, H, , OH, , H, H, , 0, , H, , O, H, , OH OH
Page 97 :
Lactose (β-D-galactopyranosyl-β-D-glucopyranose), Lactose is called milk sugar. The glycosidic linkage is β- (1,4), glycosidic linkage., It is hydrolysed by the enzyme emulsin., , OH, OH, H, HO, , H, H, , OH, , H, , H, , O, , OH H, , 0, , H, , HO, H, , H, , O, H, OH OH
Page 98 :
Determination of ring size in glucose, Methods of ring size determination in glucose, , 1) Hudson’s Periodic acid oxidation method-Periodate, method or Periodic acid (HIO4) oxidation, 2) Exhaustive methylation followed by Fischer, Concentrated Nitric acid Oxidation method
Page 99 :
Hudson’s Periodic acid oxidation method-Periodate method, , Hudson used periodic acid (HIO4) to oxidise D-methylglucoside., It specifically oxidises a glycol group to two carbonyl groups., , It is an excellent method of proving the presence of a six, membered ring in glucose
Page 101 :
Glucose is converted to methyl glucoside to eliminate the, complication of oxidation of the open chain aldehydic form and then, treated with excess of periodic acid., Two molecules of periodic acid are used for oxidation of methyl, glucoside., One molecule of acid is used for each pair of adjacent alcoholic, groups., Further oxidation of the product obtained and subsquent hydrolysis, yields optically active dihydroxypropionic acid., This establishes the size of the ring as six membered.
Page 102 :
If glucose formed a five membered ring, one molecule of, formaldehyde would have been formed instead of formic, acid., Also the major fragment would be a trialdehyde., This trialdehyde on oxidation with bromine water, followed by hydrolysis yield hydroxy malonic acid., Thus dihydroxy propionic acid cannot be obtained from, the furanose form., , So D-(+)-glucose has a pyranose ring structure and ring, has the chair form.
Page 108 :
From the above sequence of reactions the free -OH group, indicates which carbon is involved in the ring formation., , On concentrated nitric acid oxidation the –CHO is oxidized, to -COOH group and free -OH group to keto group., The keto group is susceptible to further oxidation on, concentrated Nitric acid to give a mixture of two diacids C4, acid and C5 acid., It is possible only if the cleavage is on either side of C5., , This means that -OH should be at C5 and it is C5 which is, involved in the ring formation., This means that the ring must be six membered.
Page 109 :
If methyl-α-D-glucoside had contained a five membered, ring the electron sequence described above would have led, to the formation of dimethoxy succinic acid and acids of, smaller number of carbon atoms.