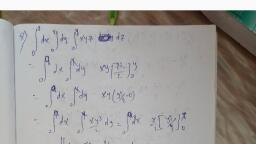Page 2 :
fagnetic Dipole Moments, Electron Spin and Vector Atom Model, , ie addition of orbital and spin angular momentum vectors. This leads to the vector model, , the atom., a Let us consider an atom whose total angular momentum is provided by a sine, , electron*. The magnitude of the orbital angular momentum i of an atomic electron is, , given by, = ee, 2, , and its z-componentis, , h, L=ma7, ~ Where J is orbital quantum number and my is the corresponding magnetic quantum, number, with values, my = b= 1,...0...,78 + Lok, Similarly, the magnitude of the spin angular momentum Sis given by, S= +, Set) aa one, , and its z-component is, , S,= Do, , where s is the spin eC number (which has the sole value + 3) and m, is magnetic spin, quantum number (7m, = +i = 255),, , >., , The total angular momentum of the one-electron atom, J, is the vector sum of, > Pines, L and §, that is, , , , , , , , , , , , , , , P=L+8., , : > aa ule, The magnitude and the z-component of J are specified by two quantum numbers j, m,. According to the usual quantisation conditions «, , J=VjG+) *, and Is 1, J is called the ‘inner quantum number’ and my is the corresponding wo, , number, The possible valucs Of, range” Uj from +j to —/ in integral steps |, TP =F, J sly os. O cataray tds:, , ss us obtain the relationship among the various angular momentum q, ince J,,L, and S, are scalar quantities, we He write, , This gives, , aS, i, Sey, It, z 5, , , , , , *Atoms of H, Li, N, la, etc are of this kind,, (except for hydrogen which has no inner fons), the, , Zero. lons He*, Be, Ma", Bt, alt ete als ‘belong to this
Page 3 :
Fr * Atomic and Molecular Spe., . :, , , , , , , , , , , , , , , , , ig? esp i :, ‘Since m) is an integer and my ist 3" so mj must be half-integral. The ma:, , J ands respectively. Therefore, from the last expression, \, Jetty., , , , of mm and m, are j,, , , , , , , , Like mj, j 1s always half-integral., , >. i :, Since 7 L and S are all quantised, they can have only certain specific relat, F : t), orientations. In case of a one-electron atom, there are only two relative orienta, possible, corresponding to, , pelts, so that Loe 4, and Gimoliorss so that Tiel :, The two ways in which L and S’can combine to form J (when l=1,s=!, , = 1, 5 =>) are shown in, , Fig. 5., , S, , oy, , Sy, i : nium J is i, Magnitude and direction. Cae Conserved in, , os nvariant. From the cosine, , , , PA att) ti, , z ae oo 21 Scos (LS), Peutos (LS). J 2 2g? aa, an bs, , , , pele ice