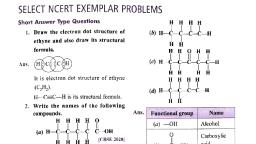
Page 1 :
IO meen rant psncseeetermn sneer ONT eI « > DP er, Ey S pradecpe Dervnene, | s series differ by, of a homologous, 2. The molecular formulae of any two successive (adjacent) members of, a CH) unit. For example 4 Hatt, , - u, ‘ iffer by a CF, CH, and C,H¢ ae the first and the second member di tel ae, C,H, and C fe ber differ by < 2, 20k. H ae » second and the third mem ;, gaat the second and t CH, unit, , C3Hg and CyHjo — the third and the fourth member differ by 4, , imi ‘ ‘ 4 s of ho, ____Insimilar way, we can explain that any other two successive members, differ by a CH, unit., 3. The molecular masses of any two successive mem, , : For example, the molecular mass of CH, is 16 u (the atomic mass, ydrogen is 1 u, therefore, molecular mass of CH, = 12 x 1 +4 1 = 16 u) an, : : 4 ss imilarly, an sh, , +6 x 1 = 30u). Thus, the difference in their molecular masses is = 30 — 16= 14 u. Stee wec Ow, that the difference in molecular masses of any other two successive members is always d, , 4. All the members of a given homologous series have the same functional STOUP: For eee pes, functional group of alcohols is the hydroxyl, i.e., —OH group. Similarly, the functional group Of aldenydes, is the aldehydic group, i.e., —CHO and that of the ketones is the ketonic group, 1-@., >C = 0., , 5. All the members of a homologous series show similar chemical properties. For example, all the, alkanes, i.e., methane, ethane, propane, etc. undergo substitution reactions with halogens (i.e., fluorine,, chlorine, bromine, iodine, etc.). Thus,, , mologous series of alkanes, , bers of a homologous series differ by 14 u., of carbon = 12 u and that of, d that of ethane is 30 u (2 x12, , igh, CH ec eee CHC + HCI, , Methane Chlorine Chloromethane Hydrogen chloride, 6. The members of a homologous series show a gradation in physical properties as the molecular, mass increases. For example, the melting points and boiling points increase with increasing molecular, mass. Other physical properties like solubility in a particular solvent also show a similar gradation., , 4.8. NOMENCLATURE OF CARBON COMPOUNDS, , Many organic compounds have two names :, , (i) Trivial or common names. These names were given after the source from which the organic, compounds were first isolated. For example, acetic acid got its name from acetum (Latin : acetum means, vinegar) since it is present in vinegar. Similarly, formic acid (methanoic acid) got its name from formicus, (Latin : formicus means red ant). Since it was first obtained by distillation of red ants. Likewise methyl, alcohol (methanol) was called Wood Spirit because it was first obtained by destructive (in absence of air), distillation of wood., , (ii) IUPAC names. International Union of Pure and Applied Chemistry (TUPAC) have given certain, rules to systematize the nomenclature of organic compounds. The names based upon these rules are called, TUPAC names and are most widely used. The IUPAC name of any organic compound can be derived by, using the following rules :, , 1. Identify the number of carbon atoms in the compound., , A compound containing one carbon atom will be named after methane., , A compound containing two carbon atoms will be named after ethane., , A compound containing three carbon atoms will be named after propane., A compound containing four carbon atoms will be named after butane., A compound containing five carbon atoms will be named after pentane., A compound containing six carbon atoms will be named after hexane.