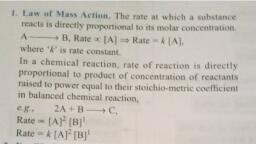Page 1 :
CHEMICAL EQUILIBRIUM, , , , INTRODUCTION, , @), , ), , ©, , @, , Whenever we hear the word Equilibrium immediately a picture arises in our mind an object under the, influence of two opposing forces. For chemical reactions also this is truc. A reaction also can exist ina, state of equilibrium balancing forward and backward reactions., , Symbolic representation of any chemical change in terms of reactants and products is called chemical, reaction., , Types of chemical reaction :, On the basis of physical state, , , , Homogeneous reactions, , Allreactants and products are in same phase, N,(g) + 3H,(g) = 2NH,(g), , Heterogeneous reaction, , Reactants and products are in two or more phase}, Zn(s) + CO,(g) = ZnO(s) + CO(g), , , , On the basis of direction, , Reversible reaction, , Irreversible reaction, , , , , , , , @ Chemical reaction in which products Chemical reaction in which products cannot, can be converted back into reactants be convert back into reactants, H, +1, = 2HI Zn + H,SO, - ZnSO, + H,, , @ Proceed in forward as well as backward Proceed only in forward direction, direction, , (a) ~— Theseattain equilibrium These do notattain equilibrium, , (iv) _ Reactant arenever completely Reactants are nearly completely converted, converted into products into products, , (v) Generally thermal dissociations are held in Generally thermal decompositions are held, closed vessel in open vessel, PCI,(g) = PCI,(g) + Cl(g) 2KCIO,(s) > 2KCI(s) + 30,(g), , On the basis of speed, Fast reactions Slow reactions, , @ Generally these reactions are ionic Generally these reactions are molecular, in nature innature, HCI + NaOH — NaCl + H,O H, +1, 2HI, Acid Base Salt Water, , On the basis of heat, Exothenmic reaction Endothermic reaction, , @ Heat is evolved in these type of chemical Heat is absorbed in these type of chemical, , reactions, R—P+xkcal, , , , reactions, R—P-x kcal
Page 2 :
Itis an experimental fact that most of the process including chemical reactions, when carried out ina, closed vessel, do not go to completion. Under these conditions, a process starts by itself or by initiation,, continues for some time at diminishing rate and ultimately appears to stop. The reactants may still be, present but they do not appear to change into products any more., , EQUILIBRIUM AND ITS DYNAMIC NATURE, , q), , Q), , 8), , Definition : "Equilibrium is the state at which the concentration of reactants and products donot change, with time. i.c. concentrations of reactants and products become constant.”, , Characteristics : Following are the important characteristics of equilibrium state,, , w, , @, , @, (w), , ), , Types:, , @, @, @), , (w), , Products, , Concentration —>, , , , Equilibrium state can be recognised by the constancy of certain measurable properties such as, pressure, density, colour, concentration etc. by changing these conditions of the system, we can, control the extent to which a reaction proceeds., , Equilibrium state can only be achieved in close vessel, but if the process is carried out in an open, vessel equilibrium state cannot be attained because in an open vessel, the reverse process may, not take place., , Equilibrium state is reversible in nature., , Equilibrium state is also dynamic in nature. Dynamic means moving and at a microscopic level,, the system is in motion. The dynamic state of equilibrium can be compared to water tank having, an inlet and outlet. Water in tank can remain at the same level ifthe rate of flow of water from, inlet (compared to rate of forward reaction) is made equal to the rate of flow of water from, outlet (compared to rate of backward reaction). Thus, the water level in the tank remains constant,, though both the inlet and outlet of water are working all the time., , At equilibrium state, i \, , & Equiorum, Rate of forward reaction = Rate of backward reaction : [f =, , 2, , Equilibrium in a system implies the existence of the following types of equilibrium simultaneously,, Thermal equilibrium : There is no flow of heat from one part to another i.e. T = constant., Mechanical equilibrium : There is no flow of matter from one part to another i.c. P= constant., Physical equilibrium : It isa state of equilibrium between the same chemical species in different, phases (solid, liquid and gascous), , Chemical equilibrium : There is no change in composition of any part of the system with time.
Page 3 :
Physical equilibrium., , qa, , Q), , The various equilibrium which can exist in any physical system are,, , Solid = Liquid, Liquid = ‘Vapour, Solid aah Gas(vapour), Solid — Saturated solution of solid in aliquid, Gas(vapour) => Saturated solution of gas in a liquid, Solid-liquid equilibrium, , H,O(s) => H,O (liquid), , Rate of transfer of molecules from ice to water = Rate of transfer of molecules from water to ice, Rate of melting of ice = Rate of freezing of water, , Liquid-vapour equilibrium : When vapour ofa liquid exists in equilibrium with the liquid, then, Rate of vaporisation = Rate of condensation,, , H,O(liquid) => H,O(vapour), , Conditions necessary for a liquid-vapour equilibrium, , @, @, , @, , 8), , The system must be a closed systemi.e., the amount of matter in the system must remain constant., The system must be at a constant temperature., , , , , , The visible properties of the system should not change with time., , Solid-vapour equilibrium : Certain solid substances on heating get converted directly into vapour, without passing through the liquid phase. This process is called sublimation. The vapour when cooled,, gives back the solid, it is called disposition., , Solid ==> Vapour, The substances which undergo sublimation are camphor, iodine, ammonium chloride etc., For example, Ammonium chloride when heated sublimes., , Heat, , NH, Cl(solid) =r NH,Cl(vapour)
Page 4 :
@), , (5), , Equilibrium between a solid and its solution : When a saturated solution is in contact with the solid, solute, there exists a dynamic equilibrium between the solid and the solution phase., , Solid substance ==> Solution of the substance, Example : Sugar and sugar solution. Ina saturated solution, a dynamic equilibrium is established between, dissolved sugar and solid sugar., , Sugar (solid) => Sugar (aqucous), At the equilibrium state, the number of sugar molecules going into the solution from the solid sugar is, equal to the number of molecules precipitating out from the solution, i.c., at equilibrium,, Rate of dissolution of solid sugar = Rate of precipitation of sugar from the solution., , Equilibrium between a gas and its solution in a liquid : Gases dissolve in liquids. The solubility ofa, gas in any liquid depends upon the,, , (i) Nature of the gas and liquid., , (ii) Temperature of the liquid., , (iii) Pressure of the gas over the surface of the solution., , Characteristics of chemical equilibrium :, , (a) It is a dynamic equilibrium i.e. at this stage, reaction takes place in both the directions with same, speed so, there is no net change., , (&) — At equilibrium the reaction proceeds both the side, equally, , (c) At equilibrium, both reactants and products are present and their concentration do not change with, respect to time., , @ _ _—_ Thesstate of equilibrium is not effected by the presence of catalyst : It only helps to attain the, equilibrium state in less or more time., , (e) — Change in pressure, temperature or concentration favours one of the reactions and thus shifts, the equilibrium point in one direction., , RATE OF REACTION, , In areaction , there is change in concentration of reactant or product per mole in unit time, it is known, as rate of the reaction., , 7 i i dc, Rate of reaction = SG) shane ln:concontmation off seaciant Jeiengeiin ae of seoctont --( } reactant, , time a, , Here negative sign indicate that concentration of reactants decrease with time., , change in concentration of products, time, , 8) ot, , Here positive sign indicate that concentration of products increase with time., , Rate of reaction = +
Page 5 :
Note : The concentration change may be positive or negative but the rate of reaction is always positive., , mole/lit, _mole, , ii ion = => = it* “4, Unit of rate of reaction litsec mole lit"' sec, , For example A> B, , djA, For reactant — — la] (concentration decrease with time], , a[), , dt, [d [A], d [B] are change in concentration of A & B in time dt}, At equilibrium, since there is no net change in concentration of reactant or product., , For reactant — + [concentration increase with time], , So rate of reaction is zero., - la) = sl] =0(At equilibrium), , LAW OF MASS ACTION, , (a) This law was given by Guldberg and Waage., , (®) At a given temperature, the product of the concentration of products each raised to the corresponding, stoichiometric coefficients in the balanced chemical equation divided by the product of the, concentrations of the reactants raised to the corresponding stoichiometric coefficients has a constant, , value., A+B=C+D, Rate of chemical reaction r x [A][B], r =K[A][B], Mathematical Expression, , @ For unitary stoichiometeric coefficients, At the constant temperature, Ict us consider the following reversible reaction., A+B=C+D, According to law of mass action Rate of forward reaction, rp [A][B] or = K,[A] [B], where K,is the rate constant of the forward reaction.