Page 1 :
13. , AMINES
Page 2 :
Amines are the derivatives of ammonia. , Like ammonia, the nitrogen atom in amines is also sp3 hybridised with an unpaired electron in one of the sp3 hybridised orbitals. , So the shape of amines is also pyramidal., Amines are classified into three types – primary (10 ), secondary (20 ) and tertiary (30 ) amines. , If one hydrogen atom of ammonia is replaced by R (alkyl) group, we get 10 amine. Their general formula is R-NH2. , If two hydrogen atoms of ammonia are replaced by two R or Ar groups, we get 20 amine. Their general formula is R2NH. , If three hydrogen atoms of ammonia are replaced by R group, we get 30 amine. Their general formula is R3NH
Page 3 :
Preparation of Amines , Reduction of Nitriles: , Nitriles on reduction with lithium aluminium hydride (LiAlH4) or catalytic hydrogenation produce primary amines. , This reaction is used for ascending in amine series, i.e., for preparation of amines containing one carbon atom more than the starting amine.
Page 4 :
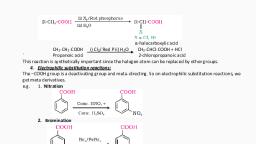
2. Reduction of amides: , The amides on reduction with lithium aluminium hydride give amines., 3. Hoffmann bromamide degradation reaction: , This method is used for the conversion of an amide to a primary amine with one carbon atom less than that present in the amide. , -In this method, an amide is treated with Bromine and ethanolic solution of NaOH to give an amine.
Page 5 :
Chemical Reactions , Carbylamine reaction (isocyanide test): , Aliphatic and aromatic primary amines on heating with chloroform and alcoholic potassium hydroxide form foul smelling isocyanides or carbylamines. , This reaction is known as carbylamines reaction or isocyanide test and is used as a test for primary amines. , Secondary and tertiary amines do not give this reaction.
Page 6 :
2. Reaction with benzene sulphonyl chloride [Hinsberg Test]: , This test is used for the distinction of primary, secondary and tertiary amines and also for the separation of a mixture of amines. Benzenesulphonyl chloride (C6H5SO2Cl) is known as Hinsberg’s reagent. , a) Primary amines react with benzenesulphonyl chloride to form a precipitate (N-alkyl benzenesulphonamide), which is soluble in alkali.
Page 7 :
b) Secondary amines react with benzene sulphonyl chloride to give a precipitate (N,N-dialkylbenzene sulphonamide), which is insoluble in alkali., c) Tertiary amines do not react with benzenesulphonyl chloride. Nowadays, Benzenesulphonyl chloride is replaced by p-toluenesulphonyl chloride
Page 8 :
AROMATIC DIAZONIUM SALTS , They have the general formula ArN2+ X – where Ar is an aryl group and X– may be Cl– , Br – , HSO4 – , BF4− , etc. The N2+ group is called diazonium group. , Preparation – Diazotisation: Aromatic diazonium salts are prepared by treating an aromatic primary amine with Nitrous acid (which is prepared by mixing NaNO2 and HCl) at 273 – 278K (0-5 0 C). , The conversion of primary aromatic amines into diazonium salts is known as diazotisation., For example benzenediazonium chloride is prepared by the reaction of aniline with nitrous acid at 273-278K
Page 10 :
Reactions involving retention of diazo group Coupling reactions:, When benzene diazonium chloride is treated with phenol or aniline, the para position of is coupled with the diazonium salt to form p-hydroxyazobenzene or p-aminoazobenzene. This type of reaction is known as coupling reaction. , This is an example of electrophilic substitution reaction.