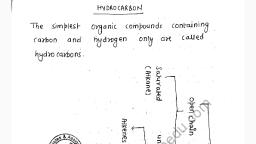
Page 1 :
Electrochemistry, , ], Electrochemistry is that branch of chemistry which deals with the study of production of, electricity from energy released during spontaneous chemical reactions and the use of electrical, energy to bring about non-spontaneous chemical transformations,, , Importance of Electrochemistry, , 1, Production of metals like Na, Mg. Ca and Al,, 2. Electroplating., , 3. Purification of metals., , 4. Batteries and cells used in various instruments., , Conductors, Substances that allow electric current to pass through them are known as conductors., Metallic Conductors or Electronic Conductors, , Substances which allow the electric current to pass through them by the movement of electrons, are called metallic conductors, ¢.g.. metals,, , Electrolytic Conductors or Electrolytes, , Substances which allow the passage of electricity through their fused state or aqueous solution, and undergo chemical decomposition are called electrolytic conductors, e.g., aqueous solution, of acids. bases and salts., , Electrolytes are of two types;, 1, Strong electrolytes The electrolytes that completely dissociate or jonise into ions are, called strong electrolytes, e,g., HCl, NaOH, K,SO,;, , 2. Weak electrolytes The electrolytes that dissociate partially (ex < 1) are called weak, electrolytes, e.g., CHx,COOH, HxCOs, NHyOHH2S, ete., , Electrochemical Cell and Electrolytic
Page 3 :
1, There is no evolution of heat., 2. The solution remains neutral on bath sides., 3. The reaction and now of electrons stops after sometime., , Daniell Cell, , An electrochemical cell of zinc and copper metals is known as Daniell cell. It is represented as, , , , Zn(s)| Zn**(ag){| Cu® (ag)| Cu(s), LHS oxidation, Zn —+ Zn** + 2e7, RHS reduction, | Cu** +2e —+ Cu, Overall reaction, Zn +Cu* (aq) —+ Zn**(ag)+Cu, , By convention cathode is represented on the RHS and anode on the LHS., Function of salt bridge, , 1. It completes the circuit and allows the flow of current., , 2. It maintains the electrical neutrality on both sides. Salt-bridge generally contains, solution of strong electrolyte such as KNOy, KCL ete. KCI is preferred because the, transport numbers of K” and Clare almost same., , Transport number or Transference number The current flowing through an electrolytic, solution ts carried by the ions. The fraction of the current carried by an ion is called its, transport number or transference number. Thus,, , Transport number of cation. n, = (current carried by cation/total current), Transport number of cation, ny = (current carried by anion/total current), Evidently ne + 05 =1, , Electrode Potential
Page 4 :
a| Page, , When an electrode is in contact with the solution of its ions in a half-cell, it has. a tendency to, lose or gain electrons which is known as electrode potential. It is expressed in volts. [t is an, intensive property, Le., independent of the amount of species in the reaction,, , Oxidation potential The tendency to lose electrons in the above case is known as oxidation, potential, Oxidation potential of a half-cell is inversely proportional to the concentration of ions, in the solution,, , Reduction potential The tendency to gain electrons in the above case is known as reductic., potential. According to TUPAC convention, the reduction potential alone be called as the, electrode potential unless it is specifically mentioned., , Et pam FE cxtdutban, , It is not possible to determine the absolute value of electrode potential. For this a reference, electrode [NHE or SHE] Is required. The electrode potential is only the difference of potentials, between two electrodes that we can measure by combining them to give a complete cell., , Standard electrode potential The potential difference developed between metal electrode and, solution of ions of unit molarity (1M) at 1 atm pressure and 25°C (298 K) is called standard, electrode potential,, , It is denoted by E°., Reference Electrode, , The electrode of known potential is called reference electrode. It may be primary reference, electrode like hydrogen electrode or secondary reference electrode like calomel electrode., , Standard hydrogen electrode (SHE) Standard hydrogen electrode (SHE), also known as normal, hydrogen electrode (NHE), consists of platinum wire, carrying platinum foil coated with finely, divided platinum black. The wire is sealed into a glass tube, placed in beaker containing 1 M, HCL. The hydrogen gas at | atm pressure is bubbled through the solution at 298K. Half-cell is, pt Hy (1 atm) H” (1 M), , Connecting wire, 1M HCI solution, , . . “F Platinum foil, , coated with Pt, black
Page 5 :
S| Page, , In SHE, at the surface of plantinum, either of (he following reaction can take place, 2H" (ag) + 2e —» HyG Reduction, , Hyg) > 2H (ag) + Ze Oxidation, , The electrode potential of SHE has been fixed as zero at all temperatures., , Its main drawbacks are, , 1, Itis difficult to maintain 1 atm pressure of H» gas,, 2, Itis difficult to maintain H” jon concentration | M., 3. The platinum electrode Is easily poisoned by traces of impurities., , Hence, calomel electrodes are conveniently used as reference electrodes, It consists of mercury, in contact with Mge Cly (calomel) paste in a solution of KCI., , Electromotive Force (emf) of a Cell, , It is the difference between the electrode potentials of two half-cells and cause flaw of current, from electrode at higher potential to electrode at lower potential, It is also the measure of free, energy change. Standard emf of a cell,, , Foy = Exitos ~ Eenote = Erg ~ Eien = Eret + Bos, , Potential differance between two The potential difference of tho two halt, , oloctrodes when no cument is flowing | cells when eléctric currant Nows, Ip Oe chreuit Is called aenif, e through the calls © called call potential., , 2. | Bret is the rraximum voltage which car | It is ohvays Jess than the maximum, be obtained trom the call, Vottage obtainable trom the cai, , 3 | Ent ts measured by 2 potentiometer, — | It ls measured by @ vollmeter,, , , , ee eed, , , , , , , , , , , , , , , , Electrochemical Series, It is the arrangement of electrodes in the increasing order of their standard reduction potentials,, , Standard Electrode Potential at 298 K