Attend Live Classes using Any Device be it Phone, Tablet or Computer
Get Started Today
Experience Teachmint X - AI driven Interactive Flat Panels and Smart Boards
Click HereExperience Teachmint X - AI driven Interactive Flat Panels and Smart Boards
Click Here






















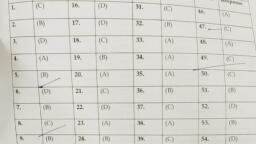








































Top Leader by G2

Top Performer by SourceForge

Top Leader by SoftwareSuggest

Ranked Amongst Top 25 Companies by LinkedIn

ISO27001 Certified

Most Preferred Workplace

We use cookies to enhance site navigation and analyse usage, read our Privacy Policy for more.


