Page 1 :
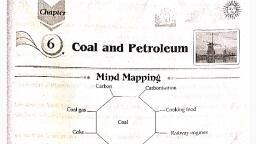
CARBON, Carbon is ranked as the third most important element next to, oxygen and hydrogen, for, the survival of life on the earth. Although it constitutes, 0.03% of the earth's crust. Because, only, of its catenation (interlinking) property, it forms innumerable, compounds with hydrogen and, oxygen. Carbon exhibits the phenomenon of allotropy. Various, allotropic forms of carbon are, listedbelow:, , CARBON, Micro-Crystalline fom (Amorphous fom), Coal, , Lamp Bleck, tis formed, , tisa, black, , by buning, , Maiena, , carbon rich, , Compounds, , vegetable, , in a, , ongn, formed by, carbonisation, under the, , eartr's surface., There are four, , types of coal, , Gas carbon, , Supply, , electrodes., , air. It is, used as a, , printer's, ink., , Fullerene, , during the, distilation, of coal., , carbon), , making gun, , Used for, , powder.It, , decolorising, , 95% carbon), Coal is used as a fuel., , America and, Siberia, it, , of graphite, , PROPERTIES, , SI02+3C-SiC+2C0, It is a dark, , conductor, , Wood charcoal Animal charcoal, Sugar Charcoal, prepared by Itis formed by C12H22011+ 11 H2804, heating wood in a, air. It is used in, , 3. Bituminus (75%, to 80% carbon), 4. Anthracite (90 to, , SIc 3500si C(Graphite), , of heat and, , t is, , limited supphy of, , 2. Lignite (70%, , by, , grey soft, , carbon), , acts as a, , reducing agent, , Diamond, In S. Africa, s., , solid which, is a good, , destructive, distillation of, bones. It is, , 1.Peat(60%, , Graphite, , PREPARATION, , destructive evaporation, , coal and, , used in making, , Coke, It is formed, , C6o, as a residue, It is formed, , during the, destructive, disbilation of, , limited, of, , Charocoal, , tis formed, , Crystalline fom, , electricity, , 12C+11H2804H20, , Upon buming, , Sugar Charcoal, , in air, it, gives carbon, , dioxide., , STRUCTURE, , organic, , compounds., , Occurs in had, rocks. In Inda, it is found in MP, and Kamataka, , PROPERTIES, Diamond is the, purest, fom of carbon, it, is the hardest, substance known, with high density, and high melting, point It has a, , In graphite, carbon, , very high refractive, , is sp hybridized., It forrms sheets, , STRUCTURE, , of hexagonal rings., one electron, is free., , In it,, , index., In diamond, carbon, , is spi hybridized. In, it, each carban is, tetrahedraly inked, with other carbon, atoms and they form, a rigid 3 dimensional, structure., , DIFFERENT FORMs OF CARBON, (1) Allotropic forms of carbon: CarbonLamp black,, , exhibits, , forms, , allotropy and occurs in the following, , (i) Diamond, , Cii) Graphite, (iii) Amorphous carbon, The other different amorphous varieties, , ofcarbon are Coal, Coke, Charcoal or wood, charcoal, Bone black is Animal Charcoal,, , Carbon black,, , Petroleum coke., , Gas carbon, , and,, , Diamond, At, , one, , time India, , was famous for, diamonds, the PiH (136.25 carat) were mined, , in India., , Properties of diamonds, , (i) In the diamond crystal, each, carbon, atom is bonded covalently to each of the, four
Page 2 :
around it, tetrahedrally. It is the densest, and purest, variety of carbon (density= 3.51)due to rather, bond distances in, short carbon-carbon, , other carbon atoms, , situated, , cal liquor and coal tar (sources of a large, number of organic compounds e.g., dyes, ex, , plosives, chemical)., , (iii) It is used in the manufacture of syn-, , diamond., , thetic, , (i) In the natural form, they do not have, shape, these are transparent to light and x, refractive index., rays and possess high, (ii) Diamonds are insoluble in all sol-, , coal., , vents. These, , are, , bad conductors of heat and, , electricity., Graphite, , Large quantities of graphite, , manufactured from coke, , or, , are, , anthracite in, , Manufacture of graphite: Artificial, graphite is now manufactured in large quantities by a process devised by Edward G., Acheson. The process consists of heating coke, anthracite coal with, , a, , little iron oxide, , or, , silica (catalyst) in a resistance type electric, , furnace., , Properties of Graphites, ) It is a dark grey substance with, charastic greasy feel and metallic lustre., (i) It marks black like lead on white, conductor of heat and, and is, , good, , paper, , electricity., , ii) Chemically it is rather, , inactive and, , somewhat dif+cult to ignite., Amorphous Carbon, , It is, , a, , of, , Coke: When coal is subjected to destruc, , absence of, , distillation by heating in the, air, it loses several volatile constituents, e.g.,, , tive, , ammonia, benzene, phenol and tar, and the residue left behind is coke. It contains, all the minerál matter of coal but is composed, coal gas,, , offree carbon primarily., , Coke is a pure variety of carbon (80 to 95, , per cent carbon), , electricfurnaces., , or, , petrol by catalytie hydrogenation, , black residue left when carbon, , compounds are heated., , Coal: Coal is believed to have been, formed by the slow carbonization of vegetable, earth centuries, matter buried underneath the, of air under high, ago. The limited supply, there, temperature and pressure prevailing, resulted in the formation of coal., The different varieties of coal available, and Anthracite, , Peat, lignite, Bituminous, VLZ.,, are different stages in the process of carbonization. The common variety of coal is, bituminous. It is hard like stone and burns, with smoky flame in anthracite., Uses of Coal, i) Coal is mainly used as a fuel., , Gi) It is used for manufacturing coal gas, and various by-products-coke and ammoni-, , Uses of Coke, (i) As a reducing agent in the iron and, , steel industry., (ii) It burns without smoke andis used as, a fuel., , (ii) For making water gas and graphite., , Oxides of Carbon, the oxides, There are two main forms ofand carbon, of carbon: Carbon dioxide (C02), mono-oxide (CO)., , Carbon Monoxide: It is an odurless, tas, teless and colourless gas. It is an extremely, , dangerous and air pollutant., Carbon monoxide is produced when, graphite is heated or burned in a limited, amount of oxygen. The reaction of steam with, red hot coke also produces carbon monoxide, along with hydrogen gas (H2)., 2C+O2»2C0, , In the laboratory, carbon monoxide is, prepared by heating formic acid (HCOOH) or, , Oxalic acid (H2C204) with concentrated sul, furic acid (H2S04)., , The mixture of carbon monoxide (CO), , and Hydrogen (H2) is called water gas and is, , used as an industrial fuel., It is useful as gaseous fuel and as a, , metallurgical agent. because at high temperatures, it reduces many metal oxides to the, elemental metal., lt is a dangerous poison. It binds to the, , haemoglobin in blood to form a compoundthat, is so stable that cannot be broken down by, body processes. When the haemoglobin is
Page 3 :
combined with carbon monoxide. It cannot, combine with oxygen, this destroys the ability, of haemoglobin to carry essential oxygen to all, parts of the body and we feel Buffocation., , CARBON DIOXIDE, , Carbon dioxide occurs in the free as well, as in combined state. In the free state, carbon, dioxide is present in atmosphere to the extent, of0.03-0.05 per cent. In the combined state, it, metallic carbonates and bicarin the earth's crust as mineral e.g.,, , occurs as, , bonates, , limestone (CaCOs), magnesite (MgCO3) and, dolomite, , (MgCO3.CaCO3) fro preparing., , but changes to gaseous state directly (i.e.,, Bublimes). Dry ice is used as a refrigerant., , Chemical Properties of Carbon Dioxide, (1)Action with litmus: It turns moist blue, litmus paper red. Thus, carbon dioxide is, acidic in nature., , CO2 +HgO, , (ii) Combustibility : Carbon dioxide is, incombustible and does not support combus, tion. Certain active metals such as mag, nesium, sodium and potassium continue, burning in a jar of the CO2 gas., , 2Mg+CO, , General Methods of Carbon Dioxide, i) By burning carbon and carbon compounds in air or oxygen completely., , C+02, , CaH4+3O2, , C021, , 2H20+2002T, , (ii) Thermal decomposition of metal carbonates and bicarbonates., , CaCO3 Ca0+CO2 1, 2NaHCO3, , Na2COs +H20, , +CO2T, (i) By the action of dil. mineral acids on, metal carbonates and bicarbonates, , NagCO3+ H2SO4, , NaHCO3 +HCl, , NagS04, , +Ha0, , +CO2, tion, , HCl, , Laboratory method for the preparaof carbon dioxide: By the action of dil., on, , marble (calcium carbonate), , CaCO3 +2HCI, , CaCl2 + H20, , +CO2T, , Carbon dioxide is collected by the upward, displacement of air because it is heavier than, air., , Physical Properties of Carbon Dioxide, 1) It is a colourless and odourless gas., (ii) It is about 1.5 times heavier than air., , ii) It is fairly soluble in water., (iv) It can be liquefied under a pressure, of 70 atmospheres at ordinary, temperature., On cooling to 78'C, it solidifies to form a, -, , white solid called dry ice which does not melt, , 2MgO+C, , 4Na+3C02, , 2NazCO3 +C, , ii) Action with lime water: Lime water, turns milky when carbon dioxide is passed, through it., , Ca(OH)+CO2- > CaCO3 +H20, Milky, The milkiness produced disappears, due, formation of calcium bicarbonate when, excess of carbon dioxide is passed., , to the, , CaCOs+H20+ CO2, , Ca(HcOa)2, , iv) Action with coke : Carbon dioxide is, , reduced, , tó carbon monoxide when, , through red hot coke at 900'C., , +CO2T, NaCl + H20, , iv), , H2COa, , passed, , . 900°C, , CO2+C, , 2CO, , (v) Photosynthesis : Plants absorb carbon, dioxide in the presence of green colouring, , matter of, , plant (chlorophyll) and sunlight to, form glucose, starch and cellulose. This, process is called photosynthesis., 6CO2+12H20- > CsH1206+6H20, , (vi)Reaction with ammonia:, , 2NH3+CO2, , +602, , (NHg)2CO+ H20, Urea, , Uses of Carbon Dioxide, 1. It is used in the manufacture of urea., 2. It is also used in the preparation o, aerated water i.e., soda water., 3. As a fire, extinguisher, 4. In the manufacture of, washing soda by, , Solvay process.