Page 1 :
ROCKS, MINERALS AND METALLLURGY, Rocks The crust of earth, , ROCKS, is made up of, igneous rocks are called volcanic or extrusive, , rocks of various types. The term 'rock' refers, , not only to hard materials like granite, but, also to soft and loose particles like sand, silt, and clay. Arock can be defined as an aggregate, of materials that form a more or less definite, unit of the ithosphere., A, , granite, but also to soft and loose particles like, , sand, silt and clay. Rocks are made up of, minerals in different combinations. Minerals, are naturally occurring solid. inorganic substances having definite chemical, composition, and physical properties. Arock can be defined, as an aggregate of minerals that forms a more, , less definite unit of the, , lithosphere., , Rocks differ from each other in colour,, texture or origin. On the basis of their mode, rocks, classified into three, types: (1) lgneous, (ii) Sedimentary, and, , can be, , Metamorphic rocks., , (ii), , 1. Igneous Rocks, The term igneous comes from the Latin, , Word, , stantly wearing down the rocks on the earth's, surface into smaller fragments. These smaller, pieces or debris are called sediments. Sediments are of various sizes, they may be gravel,, , sand, clay or very fine silt. When this sedi-, , is transported by rivers and deposited, on the bed of oceans, seas or lakes layer after, , ment, , layer for a long period, they get compacted and, , cemented together and form sedimentary, rocks. Thus, we can say that the sedimentary, rocks are formed in layers from the raw, material provided by the processes of weathe, , ring, erosion, transportation and deposition., , Classification of Rocks, , offormation,, , The word sedimentary is derived from a, Latin word 'sedimentum'which means, setting, down. Agents of gradation such as running, water, moving ice, wind and waves are con-, , rock is any large, , mass of the harder, portions ofthe earth's crust. The crust of earth, is made up of rocks of varioustypes. The term, rock' refers not only to hard materials like, , or, , rocks., 2. Sedimentary Rocks, , Sanskrit word 'agni' which, ignis', neans fire. Igneous rocks are formed by the, fiery molten material that originates far, below the surface of the earth, Igneous rocks, are formed, by cooling and solidification of, magma. Cooling of magma can take place on, or, , the surface or below the surface of the earth., Magma erupts during volcanic eruptions on, , reaching the earth's surface, gets solidified by, , cooling. When magma cools below the surface, , ofthe earth, such rocks are known as intrusive, ocks or primary rocks. When the cooling of, , agma occurs on the surface of the earth, the, , Sedimentary rocks cover 75% of the surface of, the continents. Sediment particles are com-, , posed of 80% shales, 12% hard stone and 8%, limestone., mestone, , 3. Metamorphic Rocks, , Metamorphic is a Greek word, which, , means change of form. Metamorphic rock is, not a new kind ofrock but it is a form-changed, , rock. Metamorphic rocks are formed when, igneous or sedimentary rocks are transformed, by the action of high temperature, pressure or, both. Metamorphism is the process which, , forms Metamorphic rocks. Metamorphism oc, curs when rocks are, subjected to intense heat,, , pressure or both. During the process of, , metamorphism, the original minerals get al, tered and new minerals are formed., Metamor, phism may be thermal or dynamic. During, this process shale may get changed to slate, and schist, limestone to marble, sandstone to, , quartzite and granite to gneiss.
Page 2 :
SOME COMMON METAMORPHIC, ROCK TYPES, , Rock Typde, Slate, Schist, , Description, Shale exposed to heat and pressure that splits into hard flat plates., Shale exposed to intense heat and pressure that shows evidence of shearing, , Quartzite Sandstone that is 'welded' by a silica cement into a very hard rock ofslid quartz., Marble, , Limestone exposed to heat and pressure, resulting in larger and more uniform crystals,, , Gneiss, , Rock resulting from the exposure ofsedimentary or intrusive igneous rocks to heat and, , pressure., MINERALS, Anatural inorganic substance, which pos- crust contains aluminium in the form of twa, sesses a definite chemical, well known minerals, bauxite (Al203,2H0, ecomposition, definite physical and chemical, is, and china clay (Al203, 2Si03, 2H20). But the, properties,, known as mineral. About 2,000, are, extraction of aluminium is more economical, minerals, , and, , known, , to man. Minerals, may be, to their use in various, , according, , 1. Metallic Minerals, groups:, , classified, , industries., , are divided into two, , 1) Ferrous group: They include minerals, like iron, manganese, chromite and, nickel., ii) Non-Ferrous group, In this, group, copper, 1lead, zinc, tungsten, aluminium, gold,, , silver, vanadium, etc. are included., 2. Non-Metallic Minerals are mica,, , steatite, asbestos and others., 3. Refractory Minerals: They, are used as, heat resistants in furnaces and mould., , Chronite, magnesite, kyanite, fireclays, sil-, , limanite, graphite,, , etc. are, , and easier from bauxite. Hence, ore of, , aluminium is bauxite. Thus, it can be con, cluded that all the ores are minerals but all the, minerals are not ores., It may be mentioned here that most of, the ores are either oxides or sulphides: Some, of the, , ores are also carbonates and halides., , Some examples of these various types of ores, , are given below, 1. Oxide ores, , 2. Sulphide, , important, , ores, , minerals of this group., 4. Fertilizer Minerals: They are used in, , 3., , natural gas and nuclear minerals are mineral, , Carbonate, , availability of the mineral may be the, , The mineral from which the metal, hindrance., can be economically and conveniently ex-, , tracted is called ore. For example, earth's, , (CuFeS2), , 4., , Limestone (CaCO3), , ores, , Siderite (FeCOs), , Halide ores, , Calamine (ZnC03), Rock salt (NaC), Horn silver (AgC), , fuels., , Every mineral or the metal cannot be, used for its extraction. In some cases, commercial economie factors while in other cases, , Magnetite (Feg04), Copper pyrities, Zîne blende (ZnS), Iron pyrities (Fe$2), , fertilizers. Gypsum, rock phosphate and, pyrite are included in this group., 5. Mineral Fuels: Coal, petroleum,, , Haematite (Fe20s), Bauxite (Al203.2H;0), , Among the, metals,, aluminium, abundantin, earth's, crust. It isisfollowea, the, the, iron, calcium, sodium,, and titanium in the potassium, magne, order of, , dance., , t, , um, , decreasing a, , METALLURGY, The process of, of metal from its, ore and refining is called metallurgy. Since,, the nature of the ore and also the propertiees, , possible to have the universal scheme, the, which, etals., c, h may be an, all, applicable tounivers, , of different metals are different, therefore, it, , metallurgical operations are:, , extraction, , is not, , the, the, However, some common steps involved
Page 3 :
1., , 2., , Preliminary treatment of the ore,, Reduction, , of the, , concentrated, , ore, , and, , 3. Refining., , Preliminary treatment of the ore, , Preliminary treatment of the ore invol, ves following steps:, (a) Crushing or grinding of the ore,, (6) Concentration and, (c) Conversion of ore into, , suitable form., , (a) Crushing orgrinding ofthe ore: When, the ore is taken out of the earth's crust, itis, , in the form of big lumps. These are broken to, smaller pieces by means of hammers or, mechanical crushers. Most commonly used, , crushers are jaw crushers. In these crushers,, there are two plates out of which one is movable and the other is stationary. The lumps of, ores are crushed between these two plates., The crushed ore is taken to a pulverising mill, , where it is crushed to a fine powder. For this,, either a stamp mill or a ball mill is used. The, ore is thus reduced to a fine powder., (b) Concentration or enrichment of ores, The metallic ores as mined from the earth's, CTust, are always associated with siliceouas, and earthy impurities called gague or matrix., , These impurities must be removed before the, ore is subjected to various metallurgical, operations. The process of removal of im-, , purities (unwantedearthy materials) from the, ore is called ore dressing or concentration., i) Hydraulic washing or Gravity separa-, , properties of the ore and the gangue. This, method can be applied when either the ore is, magnetic or the impurities are magnetic., Magnetite, an ore of iron, is enriched by, applying this method. Here, the ore particles, are attracted by the magnetic roller. Similar-, , ly, tungstates of iron (impurity) are temoved, from tin stone (an ore of tin) by this method., , (iv) Chemical separation or leaching, This method is based on differences between, the chemical properties of the ore and the, gangue. For example, bauxite is concentrated, by this process (Baeyer's process)., (c) Conversion of concentrated ore into a, , suitableform: After concentration, the ore is, , generally subjected to calcination or roasting,, according to the nature of the ore. During, calcination or roasting, the ore changes into, oxide. It is easier to obtain metals from their, oxides than from their sulphides or car-, , bonates., ing, , i) Calcination: It is the process of heatthe ore in a limited supply of air, at a, , temperature below its melting point., , The process results in the following changes, , 1. Volatile impurities are expelled., 2., , Moisture is removed., , 3. Carbonate ores change into oxide ores., Al203.2H20, , ZnCO3, Calamine, , tion : This method of concentration is based, upon the difference in the, , specific gravities of, , PbCO3, , the metallic ore and the gangue. The crushed, , ore is washed in the running stream of water, , when the lighter siliceous and earthy impurities (gangue) are swept away by the runningwater leaving behind heavy ore particles., This process is suitable for the concentration, of oxide ores like that of iron and tin., This method, (i)Froth floatation process:, s generally applied for the concentration of, the sulphide ores., This method is used to concentrate the, sulphide ores of copper, lead and zinc., , ii) Magnetic separation: This method is, based on the difference in the magnetic, , AljO3 +2H20, , Bauxite, , ZnO+CO2, », , PbO+CO2, , Anglesite, , Calcination is conducted in case of carbonate ores or hydrates of oxide ores., , (ii) Roasting: It is the process of heating, the ore in the excess supply of air, at a, temperature below its melting point., , This process results in the following, , changes:, 1. Volatile impurities are expelled., 2. Moisture is removed., 3. Sulphide ores change into oxide ores., , 2ZnS+302, , Zinc blende, , 2Zn0+2S02
Page 4 :
Difference between Roasting and Caleination, 1., , 2., , Calcination, , Roasting, , Ore is heated in the, presence of air., Used generally for, sulphide, phur dioxide gas, gas ie, is given, off. ores. Hence,, , absence of air., Ore is heated in the, carbonates and, sul- Used generally, dioxide gas and, ores. Hence,, vapour are, , 2ZnS+302-, , 2ZnO+2802, , given ofi., , ZnCOg, , 4FeSa+1102, 2Fe2Os+8S02, Volatile impurities are removed, , Alg0a,2H20, as oxides, , hydrated, water, , carbon, , ZnO +CO2, , Al203+2H20, , Moisture and organic impurities are removed., , (SO2 P2Os, AsgOs)., Reduction, Reduction is the process of obtaining me, tals from their, , compounds, generally oxides., , reactivity series are prepared by this method, The process of reduction with carbon is car, , ried, , out in a blast furnace., 3. Reduction with aluminium : Certain, , After the, treatment, the ore may, be subjected to reduction process by any one, , metal oxides such as Cr203 and MnO2 arenot, , ofthe following methods, depending upon its, , easily reduced with carbon. In such cases, , nature:, , aluminium is used as a reducing agent be, , preliminary, , 1.Reduction by heat alone: The oxides of, , cause it is more electropositive than chro, , metals which occupy lower positions in there, activity series, can be reduced by the effect of, heat alone. For example, mercury is produced, roasting its ore cinnabar, HgS in air. In this, , mium or manganese. The process of reduction, of oxides with aluminium is called alumino, thermy or thermite process. Some examples, , case, the sulphide ore first changes into oxide, which on further heating hanges into metal., 2HgS+302, 2Hg0 + 2SO2, Cinnabar, , 2HgO, , 2Hg +02, , 2. Smelting (Reduction with carbon): Me, tals present in the middle of the reactivity, , series cannot be obtained by the effect of heat, alone. They are produced by reduction of their, , compounds with some reducing agent which, is usually carbon. The process of reduction of, compounds with carbon is known as smelting., In this process, the roasted or calcined, of coke o r, o r e is mixed with suitable quantity, charcoal (which acts a s a reducing agent) and, its, is heated to a high temperature above, , melting point. During reduction,, , an, , are:, , Cr2O3 +2A1, , 3MnO2 +4Al, 2Al203, 4. Electrolytic reduction : The highly, electropositive metals (that is the metals o, cupying higher positions in the reactivity, , +3Mn, , series) such as sodium, calcium, magnesium,, , etc. cannot be obtained by chemical reduction, methods. Such metals are extracted by the, , electrolytic reduction. For obtaining these, metals, their salts are electrolysed in fused, state. The commonly used salts are haides, For example, sodium metal extracted Dy, electrolytes is molten sodium chloride. The, chlorine gas is liberated at anode whereas, sodium metal is released at cathode., , addition-, , NaCl, , is also mixed with the ore to remove, , al reagent, ore. This, the impurities still present in the, combines, addition reagent is called flux. Flux, with the impurities, , to form, , a, , fusible, , At anode :2C1, , called slag., Metals like zinc, iron, copper, nickel, tin,, o c c u r in the middle of the, lead, etc. which, , Nat+, , +, , CI, , Cl2 +2e, , Atcathode;, , product, , Flux + Impurities- Slag, , Al203 + 2Cr, , as, , Na +e, Nax 2, The overall reaction may be, repre, 2NaCl, , Electrolysis, , ented, , 2Na+ Cl2
Page 5 :
Refining of metals means purification of, , of aluminium is Bauxite (hydrated oxide, of aluminium-Al203.2H20) Extraction of, ex., aluminium from its, ore (bauxité) involves ex, trom its ore, , finingofofmetals. Some ofthe commonly used, , alumina. Finally, it is refined by electrolysis., It is light and strong when alloyed, thus, , 1. Distillation: Volatile metals like zinc, , its alloys are used very widelyinthe construc, tion of machinery and domestic appliance. It, , Refining of metals, , ore, , metals. Depending upon the nature of tthe, num, 0 heu, traction of alumina (aluminium oxide Al2O3), the nature of +ha, and the, the impurities, metal and, metal, present, different methods are applied for the, from bauxite and aluminium metal from, methods are discussed below:, , and mercury get purified by this method. The, pure metal distils over and is condensed in a, receiver. The non-volatile impurities are left, , is a good conductor of electricity and is often, , used in overhead transmission cables., , behind in the retort., , 2. Liquation : The method is used for, easily fusible metals like bismuth, tin and, lead. The crude metal is placed on the sloping, hearth of a furnace and heated mildly when, , themetal melts and flows down, leaving behind the infusible impurities which remain, sticking to the floor of the hearth., , Poling: Some impurities have greater, for, metal. When, oxygen, the molten metal is stirred with green-wooden, poles, the impurities come to the surface, get, 3., , affinity, , than for the, , oxidized and form a scum which can be, removed. Wood gases (hydrocarbons from, any oxide of the metal back to, wood), reduce, the metal. Copper and tin are purified by this, process., , 4. Electrolytic refining or Electiro-refin-, , ing: This method is based on the phenomenon, of electrolysis. Many metals like copper, silver, tin, gold, zinc, nickel and chromium are, , purified by electro-refining., With the progress of. electrolysis, the, anode becomes thinner and thinner. The more, reactive metals such as iron present in the, , Crude copper anode pass into solution, while, the less reactive metals such as gold, and sil, ver fall below an anode in the form of anode, , mud and are recovered. Thus, electro-refining, , IRON, , Haematite (Fez03, magnetite Fe3Os), pyrites (FeS2) are ores of íron, but, and, iron, , iron is mostly extracted fróm haematite. After, removingg gangue of the ore it is put in the, blast furnace along with coke and limestone, , (CaCO3) and heated upto about 1900°C. Coke, , and limestone act aa flux. Carbon of coke, reduces the ore to metallic iron which comes, 'atbottom of furnace. The gangue is left over, as calcium silicate (slag). From most of.the, molten iron steel is made. Some run into, moulds to make pig iron. The production of, iron is a continuous process., Pig, Iron:Itis crude cast iron produced, in theblast furnace and castinto pigs. Pig iron, is not malleable iron having more than 2%, carbon and other impurities. These imof pig iron, and make it brittle. Pure iron is very little, it is, reactive and oxidizes, as, to rust in moisture., , purities lower the melting point, , used,, , chemically, , Wrought Iron: It is the purest commer, cial form of iron and is almost free from carb-, , on. Wrought iron is ductile and malleable., Although, it has useful mechanical prope es, but little wrought iron is manufactured.I. n, be hammered, , and bent into desired, shape, drawn into wires., , not only purifies a metal but also helps in, , Tecovering more precious metals which are, present as impurities., ALUMINIUM, , It is the most abundant metal, comprisng 8% of the earth's crust. It occurs naturally, as, , Bauxite, Cryolite and Feldspar. The main, , COPPER, , Occurrence, The chief ores of copper are : i) Copper, pyrites, CuFeS2, (ii) Cuprite or ruby copper,, , Cu20,(ii) Copper glance, CugS, (iv) Malachite, , (green),, , Cu(OH)2.CuCOs,, , 2CuCO3.Cu(OH)2., , (v), , Azurite,
Page 6 :
PRANYOGIIASAN, , Copper is also found native in U.S.A.,, , Mexico, Russia and China., , 2FeS+3O2, , 2FeO +2802, , (ii) Smelting: The, , roasted ore, , tra, getsstrans, , Out of all these, copper pyrites is the, principal ore of copper. It is found in India, mainly in district Singbhum, Dharwar and, , ferred to a, , Matigara, Extraetion, , about 15 to 20 feet in height. It is mad, , 1. From oxide and carbonate ores : The, ore is crushed,, concentrated by gravity, process and roasted. Copper, carbonate, decomposes to form the oxide. Then the im-, , purities either volatilise or get oxidised., , CuCOsCu(OH)2, , 2CuO+CO2+H200, , Malachite, , The oxide so found gets reduced with, carbon in the reverberatory furnace., , CuO+C, , Cu+CO, , Cu20+C-, , 2Cu+CO, , Cuprite, 2. From copper, pyrites: Copper pyrites is, the chief ore of copper and, of, the, , process, extracting copper from copper pyrites involves, , the, , following steps, ( ) Concentration, , small blast furnace and he, , there with coke and (silica) in the preseno, excess of air. Modern copper blast furna, , sheet steel lined inside with refractory bric, All through, not only it is water jacketed but, also equipped with a waste gas outlet near he, top. The ore is placed on the charging floor, , from where it is shovelled into the furnace, , through a charging pipe., Ferrous oxide combines with silica and, comes to the surface in form of a slag., , FeO +SiO2, , FeSiO3, Iron silicate (slag), , Cuprous oxide also reacts with FeS again, , forming Cu2S., Cu20+FeS> Cu2S + FeO, This is the major problem while extract, ing copper because so long as any iron is, present there as an impurity, Cu20 cannot be, , Copper pyrites, , formed because the affinity between iron and, , (CuFeS2) in nature is associated with a huge, , oxygen isgreater than that between copper and, , quantity of unwanted siliceous impurities., , 0xygen., , For removing them first the ore is crushed,, sieved and concentrated by froth floatation, process., (i) Roasting : The concentrated ore is, heated strongly in'a cunrent of air on the, , The mass so formed mostly, contains, cuprous sulphide with a little FeS. It is known, as matte., , (iv) Bessemerisation The, , molten matte, , ofthis treatment, certain changes take place, , is now introduced into a Bessemer, converter, and heated with a blast of air blown through, , The changes that occur as a result ofthe above, mentioned treatment have been listed below, , verter from the side and the, , hearth of a reverberatory furnace. As a result, , (a) An áppreciable portion of the, , sulphur, present.in ore is removed as sulphur dioxide., S+O2, , S02, , (b) The arsenie and antimony present in, the ore are removed as their volatile oxides., , (c) The pyrites is converted into a mix, ture of cuprous and ferrous sulphides., , 2CuFeS2+O2, , CugS +2FeS +SO2, , (d) CugS and FeS are further partially, oxidised to ferrous and cuprous oxides., , 2CugS+ 302, , 2Cu20+2802 ..(1), , pipes called Tuyeres. The pipe enters the con, , Tuyeres are buit, , sufficiently high above the bottom. The metal, produced drops beneath the Tuyeres and, thus, escapes the oxidising action of the blast., The, converter can be tilted on a hollow, framewor, for charging and, The followng, discharging., reactions occur, in the converter, , 2CugS+302-, , 2Cu20+ 2S02, , 2FeS+302, , 2FeO+ 2S02, , F'errous, , oxide forms, , which is removed., , a, , slag with sl, , FeO+SiO2 > FeSi03, Slag
Page 7 :
When the entire lot of the iron has, noved, cuprous oxide reacts with, cuprous, , sulphide to form, , conveyed, on, , > 6Cu +SO2, , (Blister copper), The molten mass is poured out from the, onverter and allowed to cool, where upon it, up the dissolved SO2 and, , dissolve in, , below the anode, , the, of solidified, copper. The copper formed in this manner is, referred to as blister copper. It contains ap-, , produces, , on, , surface, , colour and is, =, , copper absorbs, out on cooling., sulphur dioxide which it gives, Copper is nott, Chemical: (1)Action, affected by dry air at ordinary temperature,, of carbon, but in moist air and in the presence, it is slowly converted into the green, , heat, , men-, , hearth of a reverberatory furnace when, Cu20 oxidises, which forms a, , the metal as, , dioxide,, , basic carbonate., , CuCO3.Cu(OH2, , 2Cu+H20+CO2+02, , be removed. Excess of Cu20, makes the copper brittle, it is removed by, which, , and electricity. Molten, , of air:, , impurities, e.g., iron to.FeO, while, slag with silica lining of the furnace,, a, Oxides of others escape a s gases or form, , Scum, , has a characteristicred, , malleable and ductile. It, =, , tionedbelow:, (a) Poling: The blister copper is melted, present in, , highly, , (Fig. 1.), , b. pt., has the density of 8.93, m.pt. 1083°C,, conductors of, 2320°C and is one of the best, , (v) Purification: Copper obtained as, , axygen, , anode mud., , Physical:Copper, , Au,, , above is refined by the two methods, , as, , Properties, , oroximately 2% impurities consisting of Ag,, Ni, Zn, Pb, Sn, As and S., , on the, , zine and other metals which, , cathode. Iron,, , are more, , CugS+2Cu20-, , blisters, , ions and finally deposited, , Cu, , dissolved, active than copper, a r e, in the, together with cøpper but they will stay, 8olution because their ion8 a r e less readily, and silver, reduced than copper ions. Gold, will not, which are less reactive than copper,, solution but fall to the bottom, , copper., , gradualy gves, , as, , Green copper, carbonate, , can, , On, , metal with powcovering the surface of fused, of, dered anthracite and by stirring with poles, green wood when, , reducing gases bubble, , up, , through the metal and the oxide gets reduced, to copper. The copper thus obtained, is 99.5%, , (ii) Action of non-metals : (a) Oxygen:, first red, forms, , heating (up to 1100°C) copperfurther, heating, cuprous oxide, Cu20 and o n, , (above 1100°C), it gives black cupric oxide,, , CuO, 4Cu+O2, , pure., , (b) Electrolytic refining:, cell consists, , of, , an, , The, , electrolytic, , a, , Impurecopper (anode) 6, , Cus), , 2Cu+O220u0, , thin sheet of, , Copper, , Po, , 2Cu20, , A, , anode made of blister, , cOoper, a cathode consisting of, pure copper and an electrolyte, containing, acid and sodium, Copper sulphate, sulphuric, is dissolved from the anode,, chloride., , A, , Uses, , Copper is one of the most important tech-, , nical metals., , Its chief uses, , are:, , Pure copper (cathode), , Cu ag)+2eSO44, , Cu2ag)+2e, -, , Copper sulphate solution, , Anode mud, Fig.-Electrolytic refining, , Cu), , of copper
Page 8 :
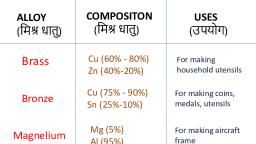
for making electrical cables and other electrical appnances., i ) Copper sheets are used for making utensils., Its alloys find, (ii) Copper forms a number of alloys like brass, bronze etc., ), , Itis used, , various, use:, arious usesi, , arts and industry., , Some important alloys of copper are given below :, , Alloys of Copper, , Alloy, Brass, , 1., , Uses, , Percentage composition, , Utensils and cartridge caps, , Copper-60%, , Zino 40%, 2. Bronze, , Control, , Copper-90%, , Medals, valves, statues, ships and, , Tin-10%, 3., , Bell metal, , Copper-80%, , Gun metal, , Tin-20%, Copper-88%, , Bells, , Gears and bearings, , Tin-10%, , Zinc-2%, ALLOYS, , An alloy may be defined as a homogeneous mixture of two or more metals or a metal anda, non-metal., , Some of the commonly used alloys, their composition, properties and uses are given in the, , following table:, Some common Alloys, their Composition,, Properties and Uses, S., No., , Alloy, Steel, , 1, 2., , 3., , Stainless steel, , Brass, , Composition, Iron (99.95%),, , Carbon (0.05%), Iron(74%),, Chromium, (18%), Nickel (8%), , Copper (80%),, Zinc (20), , Properties, , Uses, , Hard, tough and strong. Construction of ships,, bridges, vehicles etc, , Hard and does not rust., , For, , making cutlery, in-, , utensils and, , surgical, , struments, utensils, Malleable,, rong, For making, resists corrosion, can be screws, nuts and bolts., easily cast., , 4., , Bronze, , Copper (90%), Tin (10%), , Very strong and, , statues, highly| For making, making, medals, staships, , resistant to corrosion., , coins,, , propeller etc., , 5., , German silver, , (does not contain silver), , 6. Duralumin, , Copper (60%),, Zinc (20%6),, , Shines like, , silver,, , resis-, , making resistanc, | For, wires, electroplating e, , Light, strong ductile and, , For making aeroplanes, , tant to corrosion., , Nickel (20%), , Aluminium (95%), Copper in (4%), Magfxygnesium (0.5%), , resistant to corrosion., , automobile parts, pre, , sure cookers ete.
Page 9 :
49, , GENERAL SCIENCE, , 7., , Magnalium, , Aluminium (95%), Mag-| Very light and hard., , For, , nesium (5%), , ments, , making light instruand, , balance, , beams., , 8. Aluminium, bronze, , Aluminium (95%), Cop- Shineslike silver., per (5%), , For making coins, picture frames, cheapjewel-, , lery., 9, , Solder, , Lead (50%),, Tin (50%), , Has lower melting point For joining electrieal, than either lead or tin., wires together.